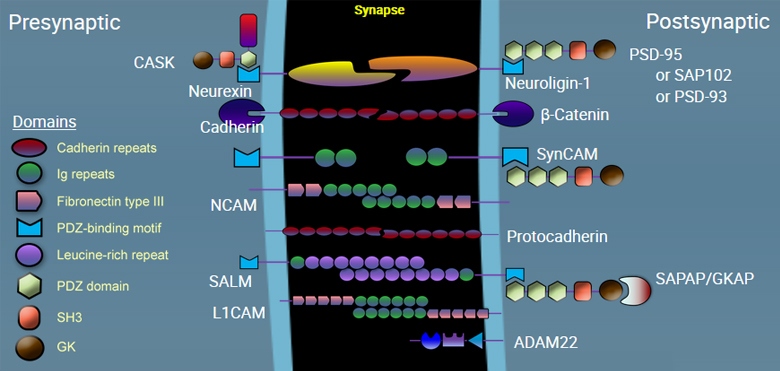
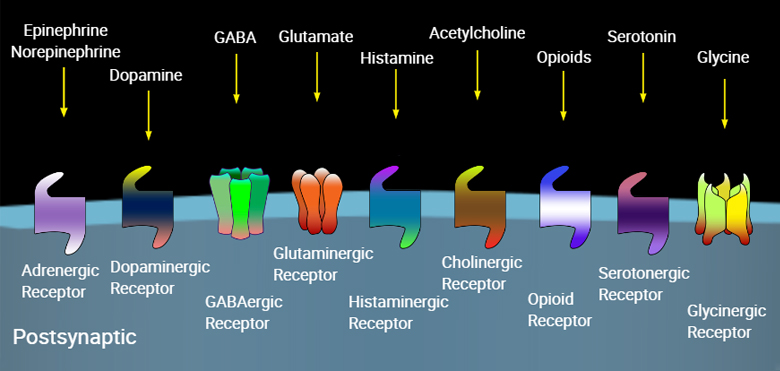
A complex array of proteins mediate the interconnection between the presynaptic membrane and the postsynaptic membrane. Adhesion molecules also contribute to a variety of processes, including the creation of synapses, mediating and modifying communication through the synapse, and modulating synaptic plasticity.
Neurexin
Neurexin is a presynaptic membrane adhesion molecule on neurons, important in the formation and specificity of synapses. It binds to neuroligin in the neuronal synapse. Within the presynaptic neuron, it associates with vesicular trafficking proteins including CASK and Mint.
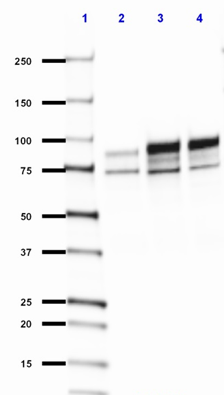
Neuroligin-1
Neuroligin is a postsynaptic membrane protein that binds to Neurexin, helping to form and maintain neuronal synapses. Within the postsynaptic neuron, it binds to proteins such as PSD-95, which recruits receptors and channels to the synapse.
View Neuroligin-1 products
Cadherin
CD325 (N-cadherin) is important for neuronal migration during development, neurite outgrowth and target finding during differentiation, initial formation of synapses, and synaptic plasticity in mature neurons. CD325 (N-cadherin) is a 130 kD single pass transmembrane protein with an extracellular region consists of five cadherin domains. N-cadherin is involved in organogenesis and in the maintenance of organ architecture by contributing to the sorting of heterogeneous cell types and in the cell adhesion needed to form tissues. N-cadherin is expressed by stem cells, myeloblasts, endothelial cells, fibroblasts, and also is expressed in neural and muscle tissues along with some types of carcinoma cells. CD325 associates with the cytoskeleton through catenin proteins.
View Cadherin products
SynCAM
SynCAM molecules mediate homotypic adhesion in the neural synapse. These proteins are also known as Necl1-4 or cell adhesion molecule 1-4.
View SynCAM products
NCAM
CD56 is a single transmembrane glycoprotein also known as NCAM (neural cell adhesion molecule), Leu-19, or NKH1. It is a member of the Ig superfamily. The 140 kD isoform is expressed on NK and NKT cells. CD56 is also expressed in the brain (cerebellum and cortex) and at neuromuscular junctions. Certain large granular lymphocyte (LGL) leukemias, small-cell lung carcinomas, neuronal derived tumors, myelomas, and myeloid leukemias also express CD56. CD56 plays a role in homophilic and heterophilic adhesion via binding to itself or heparan sulfate.
View NCAM products
Protocadherin
Protocadherins are a group of self adhering molecules belonging to the cadherin superfamily of proteins. They are predominantly expressed in neurons in widely diverse array. Unlike cadherin proteins, protocadherins do not bind to intracellular catenin molecules.
View Protocadherin products
SALM
SALM, also known as LRFN, is homotypic adhesion molecule that promotes neurite outgrowth in hippocampal neurons, regulates excitatory synapses, and induces the clustering of excitatory postsynaptic proteins, including DLG4, DLGAP1, GRIA1 (AMPA receptor subunit) and GRIN1 (NMDA receptor subunit).
L1CAM
L1CAM, also known as CD171, is a neuronal cell adhesion molecule involved in axon guidance and cell migration with a strong implication in treatment-resistant cancers. This cell adhesion molecule plays an important role in nervous system development, including neuronal migration, and differentiation. Mutations in the gene cause three X-linked neurological syndromes known by the acronym CRASH (corpus callosum hypoplasia, retardation, aphasia, spastic paraplegia and hydrocephalus). CD171 has been shown to function as a cell adhesion molecule mediating homotypic and heterotypic cell-cell interactions in neuronal myelination, neurite outgrowth and regeneration.
View L1CAM products
ADAM22
ADAM22 is a member of the ADAM (a disintegrin and metalloprotease domain) family, implicated in a variety of biological processes involving cell-cell and cell-matrix interactions, including fertilization, muscle development, and neurogenesis. Unlike other members of the ADAM protein family, the protein encoded by this gene lacks metalloprotease activity since it has no zinc-binding motif. The protein is highly expressed in the brain and may function as an integrin ligand in the brain. In mice, it has been shown to be essential for correct myelination in the peripheral nervous system.
View ADAM22 products
CASK
Calcium/Calmodulin-Dependent Serine Protein Kinase (CASK) is a multidomain scaffolding protein with a role in synaptic transmembrane protein anchoring and ion channel trafficking. It contributes to neural development and regulation of gene expression via interaction with the transcription factor TBR1. It binds to cell-surface proteins, including amyloid precursor protein, neurexins and syndecans.
View CASK products
PSD-95
PSD95, also known as SAP-90, is a member of the MAGUK family, encoded by the the DLG4 gene. At postsynaptic sites, it mediates the scaffolding of NMDA receptors, AMPA receptors, potassium channels, and other associated signaling protein clusters. It plays an important role in synaptic plasticity and the stabilization of synaptic changes during long-term potentiation.
View PSD-95 products
SAP-102
SAP-102, also known as DLG3 or neuroendocrine-DLG, is a member of the MAGUK (membrane-associated guanylate kinase) superfamily. It interacts with a number of adhesion molecules as well as glutamate receptors on the postsynaptic membrane, mediating scaffolding of synaptic signaling.
View SAP-102 products
PSD-93
PSD-93, also known as DLG2 or Chapsyn-110, is a member of the MAGUK family important in the scaffolding of receptors with adhesion molecules on the postsynaptic membrane. It regulates surface expression of receptors by directly interacting with the cytoplasmic tail of NMDA receptor subunits and inward rectifying potassium channels, and is involved in the regulation of synaptic stability at synapses.
View PSD-93 products
SAPAP
SAPAP is also known as DAP-1 or GKAP, encoded by the DLGAP1 gene. It is enriched in the postsynaptic membrane and is responsible for connecting PSD-95 and SHANK, which serves as an adapter protein that interconnects receptors of the postsynaptic membrane including NMDA-type and metabotropic glutamate receptors, and the actin-based cytoskeleton.
View SAPAP products
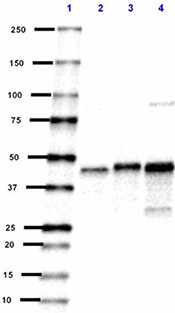
β-Catenin
β-catenin is part of a complex of proteins that constitute adherens junctions (AJs). AJs are necessary for the creation and maintenance of epithelial cell layers by regulating cell growth and adhesion between cells. The encoded protein also anchors the actin cytoskeleton and may be responsible for transmitting the contact inhibition signal that causes cells to stop dividing once the epithelial sheet is complete. β-catenin also plays a key role in Wnt signaling pathways and thus is involved in neural differentiation, synaptic plasticity, neurodegenerative disease, and prevention of apoptosis. Finally, this protein binds to the product of the adenomatous polyposis coli (APC) gene, which is mutated in the adenomatous polyposis of the colon. Mutations in this gene are a cause of colorectal cancer (CRC), pilomatrixoma (PTR), medulloblastoma (MDB), and ovarian cancer.
View β-Catenin products
 Login/Register
Login/Register 







Follow Us