- Clone
- MIH26 (See other available formats)
- Regulatory Status
- RUO
- Other Names
- BTLA, B and T lymphocyte attenuator
- Isotype
- Mouse IgG2a, κ
- Ave. Rating
- Submit a Review
- Product Citations
- publications
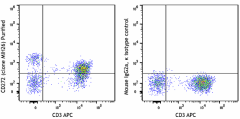
-

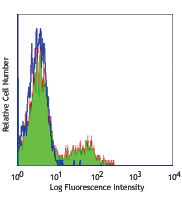
Human peripheral blood lymphocytes stained with CD3 APC and CD272 (BTLA) (clone MIH26) Purified (left) or Mouse IgG2a, κ Purified isotype control (right) followed by anti-mouse IgG FITC.
| Cat # | Size | Price | Quantity Check Availability | Save | ||
|---|---|---|---|---|---|---|
| 344529 | 100 µg | £215 | ||||
| 344530 | 1 mg | £490 | ||||
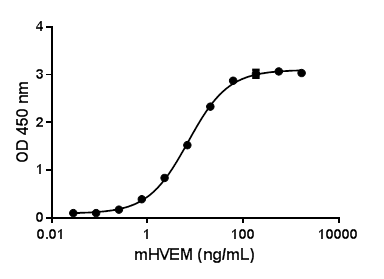
B and T lymphocyte attenuator (BTLA) is an Ig superfamily coinhibitory receptor with structural similarity to programmed cell death 1 (PD-1) and CTLA-4. BTLA is expressed on B cells, T cells, macrophages, dendritic cells, NKT cells, and NK cells. Engagement of BTLA by its ligand Herpes Virus Entry Mediator (HVEM) is critical for negatively regulating immune response. The absence of BTLA with HVEM inhibitory interactions leads to increased experimental autoimmune encephalomyelitis severity, enhanced rejection of partially mismatched allografts, an increased CD8+ memory T cell population, increased severity of colitis, and reduced effectiveness of T regulatory cells. BTLA plays an important role in the induction of peripheral tolerance of both CD4+ and CD8+ T cells in vivo. Tolerant T cells have significant up-regulated expression of BTLA compared with effector and naïve T cells. BTLA may cooperate with CTLA-4 and PD-1 to control T cell tolerance and autoimmunity. It has been reported that BTLA may regulate T cell function through binding to B7-H4.
Product DetailsProduct Details
- Verified Reactivity
- Human
- Reported Reactivity
- African Green
- Antibody Type
- Monoclonal
- Host Species
- Mouse
- Immunogen
- Human BTLA transfected cells
- Formulation
- 0.2 µm filtered in phosphate-buffered solution, pH 7.2, containing no preservative.
- Endotoxin Level
- Less than 0.01 EU/µg of the protein (< 0.001 ng/µg of the protein) as determined by the LAL test.
- Preparation
- The Ultra-LEAF™ (Low Endotoxin, Azide-Free) antibody was purified by affinity chromatography.
- Concentration
- The antibody is bottled at the concentration indicated on the vial, typically between 2 mg/mL and 3 mg/mL. Older lots may have also been bottled at 1 mg/mL. To obtain lot-specific concentration and expiration, please enter the lot number in our Certificate of Analysis online tool.
- Storage & Handling
- The antibody solution should be stored undiluted between 2°C and 8°C. This Ultra-LEAF™ solution contains no preservative; handle under aseptic conditions.
- Application
-
FC - Quality tested
Agonist - Reported in the literature, not verified in house - Recommended Usage
-
Each lot of this antibody is quality control tested by immunofluorescent staining with flow cytometric analysis. For flow cytometric staining, the suggested use of this reagent is ≤ 2.0 µg per million cells in 100 µl volume. It is recommended that the reagent be titrated for optimal performance for each application.
- Application Notes
-
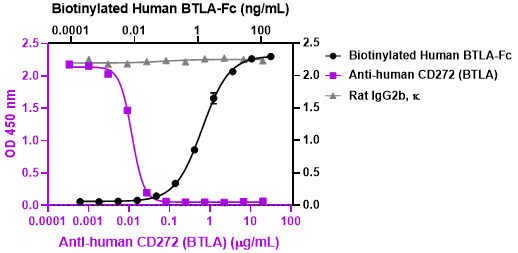
Additional reported applications (for the relevant formats) include: inhibition of T cell proliferation and cytokine production1. Clone MIH26 has agonistic activity on BTLA, resulting in the inhibition of activation.
The Ultra-LEAF™ purified antibody (Endotoxin < 0.01 EU/µg, Azide-Free, 0.2 µm filtered) is recommended for use in in vitro and in vivo biofunctional assays. -
Application References
(PubMed link indicates BioLegend citation) -
- Otsuki N, et al. 2006. Biochem. Bioph. Res. Co. 344:1121.
- Okano M, et al. 2008. Clin. Exp. Allergy 38:1891.
- RRID
-
AB_2810542 (BioLegend Cat. No. 344529)
AB_2810543 (BioLegend Cat. No. 344530)
Antigen Details
- Structure
- Ig superfamily, with similar structure to CTLA-4 and PD-1
- Distribution
-
B cells and T lymphocytes, NK cells and dendritic cells
- Function
- Negative regulation of T cell activation, proliferation and cytokine production
- Ligand/Receptor
- HVEM
- Cell Type
- B cells, Dendritic cells, NK cells, T cells, Tregs
- Biology Area
- Immunology, Inhibitory Molecules
- Molecular Family
- CD Molecules
- Antigen References
-
1. Watanabe N, et al. 2003. Nat. Immunol. 4:670.
2. Sun Y, et al. 2009. J. Immunol. 183:1946.
3. Gonzalez LC, et al. 2005. P. Natl. Acad. Sci. USA 102:1116. - Gene ID
- 151888 View all products for this Gene ID
- UniProt
- View information about CD272 on UniProt.org
Related Pages & Pathways
Pages
Related FAQs
- Do you guarantee that your antibodies are totally pathogen free?
-
BioLegend does not test for pathogens in-house aside from the GoInVivo™ product line. However, upon request, this can be tested on a custom basis with an outside, independent laboratory.
- Does BioLegend test each Ultra-LEAF™ antibody by functional assay?
-
No, BioLegend does not test Ultra-LEAF™ antibodies by functional assays unless otherwise indicated. Due to the possible complexities and variations of uses of biofunctional antibodies in different assays and because of the large product portfolio, BioLegend does not currently perform functional assays as a routine QC for the antibodies. However, we do provide references in which the antibodies were used for functional assays and we do perform QC to verify the specificity and quality of the antibody based on our strict specification criteria.
- Does BioLegend test each Ultra-LEAF™ antibody for potential pathogens?
-
No, BioLegend does not test for pathogens in-house unless otherwise indicated. However, we can recommend an outside vendor to perform this testing as needed.
- Have you tested this Ultra-LEAF™ antibody for in vivo or in vitro applications?
-
We don't test our antibodies for in vivo or in vitro applications unless otherwise indicated. Depending on the product, the TDS may describe literature supporting usage of a particular product for bioassay. It may be best to further consult the literature to find clone specific information.
Other Formats
View All CD272 Reagents Request Custom Conjugation| Description | Clone | Applications |
|---|---|---|
| Purified anti-human CD272 (BTLA) | MIH26 | FC,Agonist |
| PE anti-human CD272 (BTLA) | MIH26 | FC |
| APC anti-human CD272 (BTLA) | MIH26 | FC |
| Brilliant Violet 421™ anti-human CD272 (BTLA) | MIH26 | FC |
| PerCP/Cyanine5.5 anti-human CD272 (BTLA) | MIH26 | FC |
| APC/Cyanine7 anti-human CD272 (BTLA) | MIH26 | FC |
| PE/Cyanine7 anti-human CD272 (BTLA) | MIH26 | FC |
| Alexa Fluor® 647 anti-human CD272 (BTLA) | MIH26 | FC |
| PE/Dazzle™ 594 anti-human CD272 (BTLA) | MIH26 | FC |
| FITC anti-human CD272 (BTLA) | MIH26 | FC |
| TotalSeq™-A0170 anti-human CD272 (BTLA) | MIH26 | PG |
| TotalSeq™-C0170 anti-human CD272 (BTLA) | MIH26 | PG |
| Ultra-LEAF™ Purified anti-human CD272 (BTLA) | MIH26 | FC,Agonist |
| PE/Cyanine5 anti-human CD272 (BTLA) | MIH26 | FC |
| TotalSeq™-B0170 anti-human CD272 (BTLA) | MIH26 | PG |
| TotalSeq™-D0170 anti-human CD272 (BTLA) | MIH26 | PG |
Customers Also Purchased
Compare Data Across All Formats
This data display is provided for general comparisons between formats.
Your actual data may vary due to variations in samples, target cells, instruments and their settings, staining conditions, and other factors.
If you need assistance with selecting the best format contact our expert technical support team.
-
Purified anti-human CD272 (BTLA)

Human peripheral blood lymphocytes stained with CD3 APC and ... -
PE anti-human CD272 (BTLA)
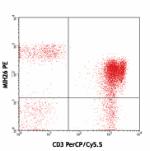
Human peripheral blood lymphocytes stained with CD3 (UCHT1) ... 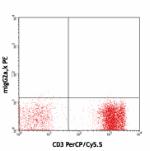
-
APC anti-human CD272 (BTLA)

Human peripheral blood lymphocytes were stained with CD19 Br... 
-
Brilliant Violet 421™ anti-human CD272 (BTLA)

Human peripheral blood lymphocytes stained with CD19 APC and... 
-
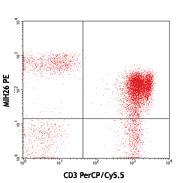
PerCP/Cyanine5.5 anti-human CD272 (BTLA)

Human peripheral blood lymphocytes stained with CD3 APC and ... -
APC/Cyanine7 anti-human CD272 (BTLA)

Human peripheral blood lymphocytes were stained with CD19 Br... -
PE/Cyanine7 anti-human CD272 (BTLA)

Human peripheral blood lymphocytes were stained with CD19 Br... -
Alexa Fluor® 647 anti-human CD272 (BTLA)

Human peripheral blood lymphocytes were stained with CD19 PE... 
-
PE/Dazzle™ 594 anti-human CD272 (BTLA)
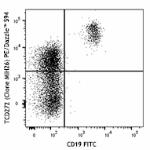
Human peripheral blood lymphocytes were stained with CD19 FI... 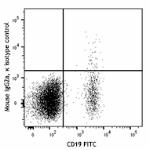
-
FITC anti-human CD272 (BTLA)
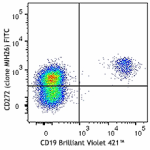
Human peripheral blood lymphocytes were stained with CD19 Br... 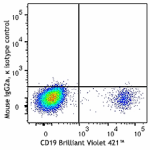
-
TotalSeq™-A0170 anti-human CD272 (BTLA)
-
TotalSeq™-C0170 anti-human CD272 (BTLA)
-
Ultra-LEAF™ Purified anti-human CD272 (BTLA)

Human peripheral blood lymphocytes stained with CD3 APC and ... -
PE/Cyanine5 anti-human CD272 (BTLA)

Human peripheral blood lymphocytes were stained with CD3 Ale... -
TotalSeq™-B0170 anti-human CD272 (BTLA)
-
TotalSeq™-D0170 anti-human CD272 (BTLA)
 Login / Register
Login / Register 









Follow Us