Tregs in Cancer and Tumor Escape
Tregs play a complex role in cancer progression. While they are essential for maintaining immune tolerance and preventing autoimmune reactions, their presence within the tumor microenvironment can have both positive and negative effects1.
Pro-Tumor Effects: In some cases, Tregs promote tumor growth and metastasis and support their survival. Tregs may facilitate the formation of new blood vessels (angiogenesis) that supply nutrients to tumors.
Tregs can also help establish an immunosuppressive environment around tumors, dampening the response of immune cells like cytotoxic T cells and natural killer cells and allowing cancer cells to evade destruction.
Anti-Tumor Effects: On the other hand, Tregs can also prevent excessive inflammation, limiting tissue damage caused by immune responses. In certain contexts, this can be beneficial for overall health.
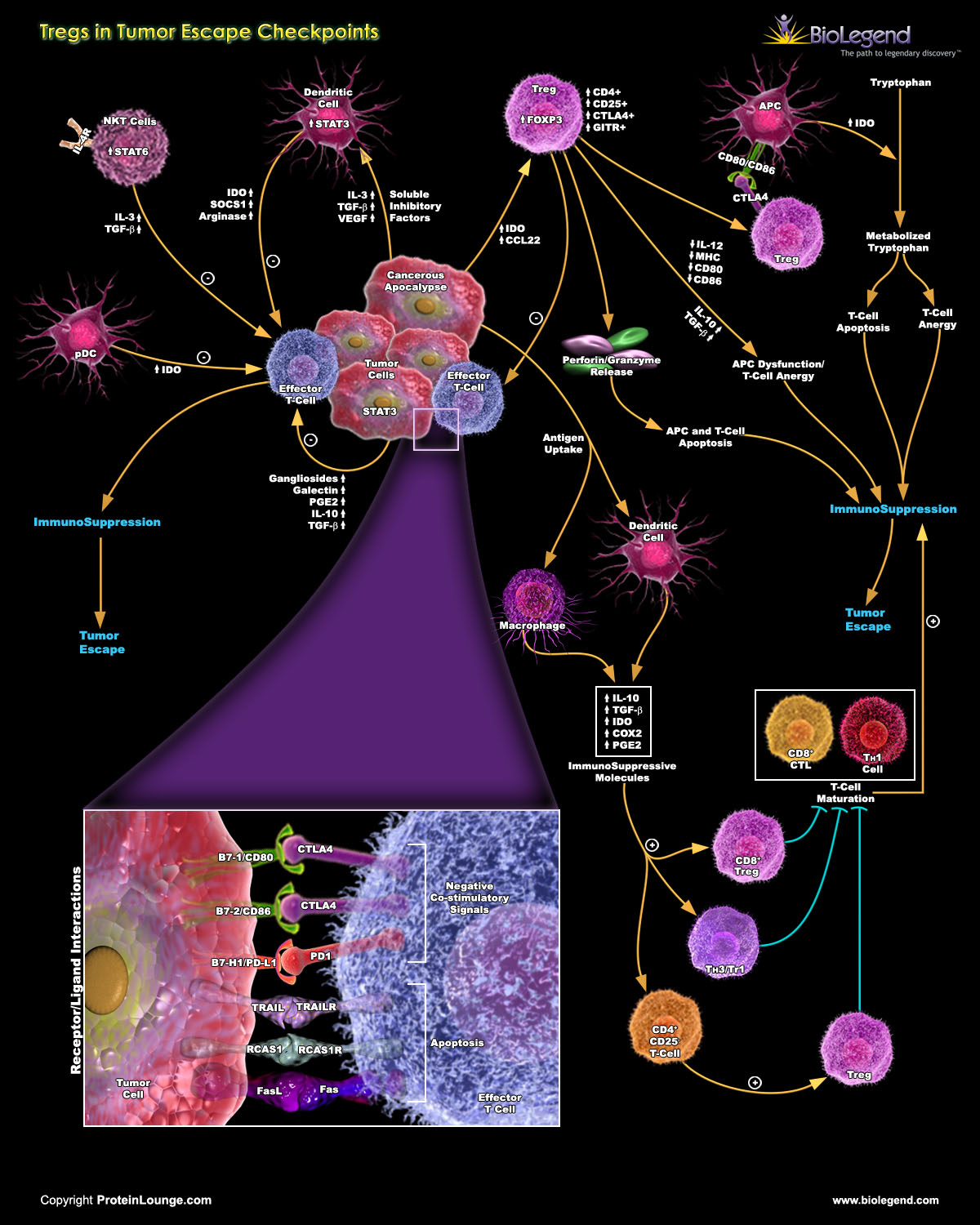
View the full pathway by visiting our webpage.
Tregs in Autoimmunity
Treg cells can suppress immune responses to self-antigens, safeguarding against autoimmunity, and have also been shown to hinder effective cancer immunotherapies. Dysregulated Treg activity has been linked to the development of several autoimmune diseases. Specifically, Treg function and frequency are often compromised in autoimmune diseases like multiple sclerosis (MS), rheumatoid arthritis (RA), or systemic lupus erythematosus (SLE)2-5.
In addition to Treg dysfunction, there’s evidence that immunoregulatory mechanisms fail to control autoreactive T cells due to reduced responsiveness of T effector cells to the suppressive effects of Tregs.
Treg cells have been shown to demonstrate plasticity, which includes the loss of FOXP3 expression. This leads to the emergence of “ex-Treg” cells and “fragile” Treg cells. Ex-Treg cells lose FOXP3 expression and may contribute to inflammatory responses, while fragile Treg cells retain FOXP3 expression but engage in an inflammatory program, compromising their suppressive activity.
A deeper understanding of Treg function could present a promising strategy for managing chronic inflammation in autoimmune disorders.
Tregs in Transplantation
In graft versus host disease (GVHD), the host immune system rejects donor lymphocytes due to differences in major histocompatibility complexes. Tregs are commonly known to induce tolerance in a host, but have also been shown as effective in limiting allogeneic responses and prevent GVHD for extended periods of time. While Tregs have shown strong potential in controlling GVHD, therapeutic approaches continue to require optimization, as Treg stability and the generation of sufficient Treg numbers for treatment remain problematic to solve6.
Treg Pathways
Visit our Pathways Library for detailed interactive pathways, including our Regulatory T Cells poster containing information on Treg development and mechanisms of action.

References
1. Hosseinalizadeh, Hamed et al. “Regulating the regulatory T cells as cell therapies in autoimmunity and cancer.” Frontiers in medicine vol. 10 1244298. 27 Sep. 2023, doi:10.3389/fmed.2023.1244298
2. La Cava, Antonio. “Natural Tregs and autoimmunity.” Frontiers in bioscience (Landmark edition) vol. 14,1 333-43. 1 Jan. 2009, doi:10.2741/3247
3. Romano, Marco et al. “Past, Present, and Future of Regulatory T Cell Therapy in Transplantation and Autoimmunity.” Frontiers in immunology vol. 10 43. 31 Jan. 2019, doi:10.3389/fimmu.2019.00043
4. Slobodin, Gleb, and Doron Rimar. “Regulatory T Cells in Systemic Sclerosis: a Comprehensive Review.” Clinical reviews in allergy & immunology vol. 52,2 (2017): 194-201. doi:10.1007/s12016-016-8563-6
5. Zhang, Yuyue et al. “Differences in Maturation Status and Immune Phenotypes of Circulating Helios+ and Helios- Tregs and Their Disrupted Correlations With Monocyte Subsets in Autoantibody-Positive T1D Individuals.” Frontiers in immunology vol. 12 628504. 12 May. 2021, doi:10.3389/fimmu.2021.628504
6. Guo, Wen-Wen et al. “Regulatory T Cells in GVHD Therapy.” Frontiers in immunology vol. 12 697854. 18 Jun. 2021, doi:10.3389/fimmu.2021.697854
 Login/Register
Login/Register 








Follow Us