- Regulatory Status
- RUO
- Other Names
- Annexin-V-Binding Buffer
- Ave. Rating
- Submit a Review
- Product Citations
- publications
| Cat # | Size | Price | Quantity Check Availability | Save | ||
|---|---|---|---|---|---|---|
| 422201 | 100 mL | $36 | ||||
Annexin V Binding Buffer has been formulated for flow cytometric labeling of apoptotic cells with Annexin V reagents. The buffer is provided as a ready-to-use solution.
Product DetailsProduct Details
- Storage & Handling
- Store between 2°C and 8°C.
- Application
-
FC - Quality tested
- Recommended Usage
-
Annexin V Binding Buffer is recommended for use with Annexin V reagents. Cells should be resuspended in Annexin V Binding Buffer at a concentration 1x106/ml and then combined with Annexin V reagents at the recommended volumes.
- Application Notes
-
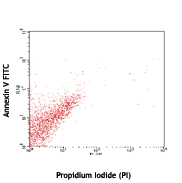
Annexin V Staining
1. Wash cells twice with cold BioLegend cell staining buffer (cat # 420201) and then resuspend cells in Annexin V Binding Buffer (cat # 422201) at a concentration of 1x106 cells/ml.
2. Transfer 100 µl of cell suspension in 5 ml test tube.
3. Add 5 µl of fluorochrome conjugated Annexin V.
4. Add 10 µl of PI solution (cat # 421301) or 7-AAD (cat # 420403/420404).
5. Gently vortex the cells and incubate for 15 min at RT (25 °C) in the dark.
6. Add 400 µl of Annexin V Binding Buffer (cat # 422201) to each tube. Analyze by flow cytometry.
For a better experience detecting apoptosis, we now recommend Apotracker™. Cell staining with Apotracker™ is Calcium independent. Thus, no special buffers are required, and the protocol can be shortened for single-step co-staining with other reagents. -
Application References
(PubMed link indicates BioLegend citation) -
- Vermes I, et al. 1995. J. Immunol. Methods 184:39.
- Soon PS, et al. 2013. Endocr Relat Cancer. 20:1. PubMed
- Turner JE, et al. 2013. J Exp Med. PubMed
- Lindner JM, et al. 2013. Mol Cell Biol. 33:4628. PubMed
- Turner JE, et al. 2013 J. Exp Med. 210:2951. PubMed
- Beggs KM, et al. 2014. Toxicol Sci. 137:91. PubMed
- Miyazawa M, et al. 2014. Mol Biol Cell. 25:2116. PubMed
- Burbulla LF, et al. 2014. Cell Death Dis. 5:1180. PubMed
- de Vires M, 2014. Am J Physiol Lung Cell Mol Physiol. 307:240. PubMed
- Sahin E, 2014. J. Immunol. 193:1717. PubMed
- Xiong R, et al. 2014. Toxicol Appl Pharmacol. 280:285. PubMed
- Thakran S, 2015. Invest Ophthalmol Vis Sci. 56:177. PubMed
- Product Citations
-
Antigen Details
- Gene ID
- NA
Related FAQs
- How is your Annexin made and what sequence does it cover?
-
It is made in E. coli, covering human aa Met1-Asp320.
- How does pH and staining temperature affect Annexin V-Phosphatidylserine binding?
-
Annexin-Phosphatidylserine binding is lost below pH 5.2 and with prolonged incubation over a temperature of 42°C.
- Why do I need to use Annexin V Binding Buffer?
-
Annexin V binding requires the presence of calcium in the solution. So, we provide Annexin V Binding Buffer (cat # 422201), which is optimized for the best performance of Annexin V staining.
- Can I use RPMI during Annexin V staining?
-
It is best to follow protocol as described on the product data sheet. Moreover, RPMI 1640 has a relatively high concentration of phosphate and low calcium ion concentration, which negatively impacts Annexin binding to its target phosphatidylserine (PS). Measurement of cell death by using Annexin V may also be significantly affected by time of incubation on ice, calcium concentration, and type of medium.
- Can I freeze Annexin V conjugates?
-
It should not be frozen as it will lead to loss of biological activity due to dimerization.
- Is Annexin V suitable for conjugation with the Maxpar® kit for CyTOF®?
-
Maxpar® Labeling kits require the protein to be partially reduced, so the metal chelate can be introduced through an SH group in the hinge region of the reduced antibody. Human Annexin V contains only one Cysteine which was reported to be chemically inactive. Thus, the Maxpar® labeling protocol would not work with Annexin V, unless a free –SH group can be introduced to Annexin V. For more information regarding SH-mediated conjugation of Annexin V please consult published papers such as this one.
Customers Also Purchased
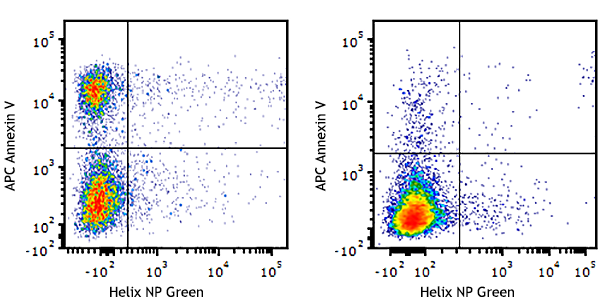
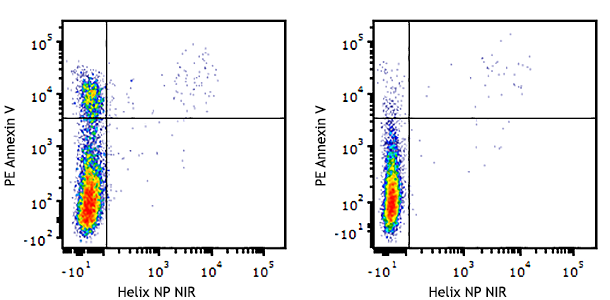
Compare Data Across All Formats
This data display is provided for general comparisons between formats.
Your actual data may vary due to variations in samples, target cells, instruments and their settings, staining conditions, and other factors.
If you need assistance with selecting the best format contact our expert technical support team.
 Login/Register
Login/Register 









Follow Us