- Regulatory Status
- RUO
- Other Names
- Hematopoietin, MVCD2, EP
- Ave. Rating
- Submit a Review
- Product Citations
- publications
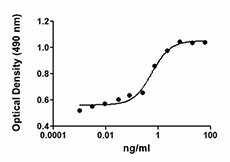
-

TF-1 cell proliferation induced by rat EPO.
| Cat # | Size | Price | Quantity Check Availability | Save | ||
|---|---|---|---|---|---|---|
| 592302 | 10 µg | £150 | ||||
| 592306 | 100 µg | £638 | ||||
Select size of product is eligible for a 40% discount! Promotion valid until September 30, 2024. Exclusions apply. To view full promotion terms and conditions or to contact your local BioLegend representative to receive a quote, visit our webpage.
Rat Erythropoietin was cloned in 1992, and it has a 79% and 95% homology with human and mouse EPOs, respectively. EPO is a glycoprotein composed of 40-60% carbohydrates, and its molecular weight varies from 30-34 kD depending on the carbohydrate percentage. Hypoxia induces erythropoiesis, and hypoxia inducible factor (HIF) is directly involved in EPO expression. HIF is a heterodimeric transcription factor (HIF-1α, HIF-2α, and beta subunits), and it is regulated by HIF-prolyl hydroxylase (HIF-PH) that hydroxylates the alpha subunits targeting them for ubiquitination and subsequent degradation. HIF-PH is a sensor of levels of iron, oxygen, and metabolic activity. High levels of HIF protein induce EPO production in the kidney and liver, and mobilization of iron to support erythropoiesis. EPO binds to EPOR, and transcripts of this receptor have been detected in non-erythroid precursors such as endothelial cells and renal proximal epithelial cells; nevertheless, it has been published that functional erythropoietin receptor is not detected in endothelial, cardiac, neuronal, and renal cells. Several transcriptions factors play a role in the expression of EPOR such as GATA1, Friend of GATA (Fog1), and the erythroid specific factor SCL/Tal1. EPO has been used to treat anemia associated with chronic kidney disease, cancer patients on chemotherapy, and as an antiviral HIV therapy.
Product DetailsProduct Details
- Source
- Rat EPO, amino acids (Ala27-Arg192) (Accession# D10763) was expressed in CHO cells.
- Molecular Mass
- The 166 amino acid recombinant protein has a predicted molecular mass of approximately 18.5 kD. The DTT-reduced and non-reduced glycosylated protein migrate at approximately 35-50 kD by SDS-PAGE. The N-terminal amino acid is Ala.
- Purity
- >95%, as determined by Coomassie stained SDS-PAGE.
- Formulation
- 0.22 µm filtered protein solution is in 1X PBS, pH 7.2.
- Endotoxin Level
- Less than 0.01 ng per µg cytokine as determined by the LAL method.
- Concentration
- 10 and 25 µg sizes are bottled at 200 µg/mL. 100 µg size and larger sizes are lot-specific and bottled at the concentration indicated on the vial. To obtain lot-specific concentration and expiration, please enter the lot number in our Certificate of Analysis online tool.
- Storage & Handling
- Unopened vial can be stored between 2°C and 8°C for up to 2 weeks, at -20°C for up to six months, or at -70°C or colder until the expiration date. For maximum results, quick spin vial prior to opening. The protein can be aliquoted and stored at -20°C or colder. Stock solutions can also be prepared at 50 - 100 µg/mL in appropriate sterile buffer, carrier protein such as 0.2 - 1% BSA or HSA can be added when preparing the stock solution. Aliquots can be stored between 2°C and 8°C for up to one week and stored at -20°C or colder for up to 3 months. Avoid repeated freeze/thaw cycles.
- Activity
- ED50 = 0.5 - 2.5 ng/ml, corresponding to a specific activity of 0.4 - 2.0 x 106 units/mg, as determined by induction of TF-1 cell proliferation.
- Application
-
Bioassay
- Application Notes
-
BioLegend carrier-free recombinant proteins provided in liquid format are shipped on blue-ice. Our comparison testing data indicates that when handled and stored as recommended, the liquid format has equal or better stability and shelf-life compared to commercially available lyophilized proteins after reconstitution. Our liquid proteins are verified in-house to maintain activity after shipping on blue ice and are backed by our 100% satisfaction guarantee. If you have any concerns, contact us at tech@biolegend.com.
- Product Citations
-
Antigen Details
- Distribution
-
EPO is primarily expressed in the kidney (70-90% of the total EPO) and liver. EPO is expressed by hepatocytes, cortical interstitial fibroblasts in ischemic kidneys, astrocytes, and neurons under hypoxia.
- Function
- EPO is the major regulator of differentiation, proliferation, and survival of erythroid progenitors. EPO expression is induced by hypoxia; HIF, among other transcription factors, regulates the EPO expression. EPO has a possible role in neuroprotection.
- Interaction
- Erythroid progenitor cells.
- Ligand/Receptor
- EpoR (homodimer).
- Cell Type
- Embryonic Stem Cells
- Biology Area
- Cell Biology, Signal Transduction, Stem Cells
- Molecular Family
- Cytokines/Chemokines, Growth Factors
- Antigen References
-
1. Nagao M, et al. 1992. Biochim. Biophys. Acta. 1171:99.
2. Chin K, et al. 2000. Brain Res. Mol. Brain Res. 81:29.
3. Snow JW and Orkin SH. et al. 2009. J. Biol. Chem. 284:29310.
4. Kassouf MT, et al. 2010. Genome Res. 20:1064.
5. Paliege A, et al. 2010. Kidney Int. (Nature) 77:312.
6. Sinclair AM, et al. 2010. Blood 115:4264.
7. Mawadda A, et al. 2012. Anatomy Research International 2012: Article ID 953264. - Gene ID
- 24335 View all products for this Gene ID
- UniProt
- View information about Erythropoietin on UniProt.org
Related FAQs
- Why choose BioLegend recombinant proteins?
-
• Each lot of product is quality-tested for bioactivity as indicated on the data sheet.
• Greater than 95% Purity or higher, tested on every lot of product.
• 100% Satisfaction Guarantee for quality performance, stability, and consistency.
• Ready-to-use liquid format saves time and reduces challenges associated with reconstitution.
• Bulk and customization available. Contact us.
• Learn more about our Recombinant Proteins. - How does the activity of your recombinant proteins compare to competitors?
-
We quality control each and every lot of recombinant protein. Not only do we check its bioactivity, but we also compare it against other commercially available recombinant proteins. We make sure each recombinant protein’s activity is at least as good as or better than the competition’s. In order to provide you with the best possible product, we ensure that our testing process is rigorous and thorough. If you’re curious and eager to make the switch to BioLegend recombinants, contact your sales representative today!
- What is the specific activity or ED50 of my recombinant protein?
-
The specific activity range of the protein is indicated on the product datasheets. Because the exact activity values on a per unit basis can largely fluctuate depending on a number of factors, including the nature of the assay, cell density, age of cells/passage number, culture media used, and end user technique, the specific activity is best defined as a range and we guarantee the specific activity of all our lots will be within the range indicated on the datasheet. Please note this only applies to recombinants labeled for use in bioassays. ELISA standard recombinant proteins are not recommended for bioassay usage as they are not tested for these applications.
- Have your recombinants been tested for stability?
-
Our testing shows that the recombinant proteins are able to withstand room temperature for a week without losing activity. In addition the recombinant proteins were also found to withstand four cycles of freeze and thaw without losing activity.
- Does specific activity of a recombinant protein vary between lots?
-
Specific activity will vary for each lot and for the type of experiment that is done to validate it, but all passed lots will have activity within the established ED50 range for the product and we guarantee that our products will have lot-to-lot consistency. Please conduct an experiment-specific validation to find the optimal ED50 for your system.
- How do you convert activity as an ED50 in ng/ml to a specific activity in Units/mg?
-
Use formula Specific activity (Units/mg) = 10^6/ ED50 (ng/mL)
 Login / Register
Login / Register 









Follow Us