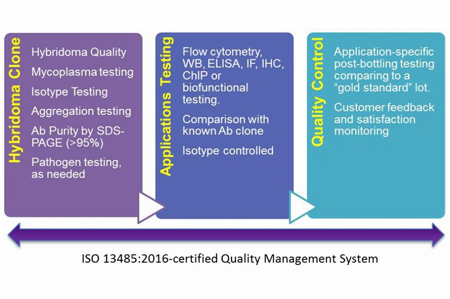
Product Development
BioLegend spends a considerable effort in developing and creating new reagents for research. We produce a variety of recombinant proteins, antibodies, immunoassays, and multiomics tools. The below example will focus on our stringency in creating new monoclonal antibodies produced from hybridomas.

Clone Development
Clones of these hybridomas are carefully selected based on a number of criteria including robust growth and efficient production of a single clone of antibody that is specific to the intended target. The best clones move on to applications testing. The steps include:
- Immunogen design and construction
- Immunization of host animal
- Hybridoma creation
- ELISA or application-specific screening of antibodies from clones
- Application testing, including WB, ELISA, ChIP, IF, IHC, or biofunctional assays
Multiple Application Validation
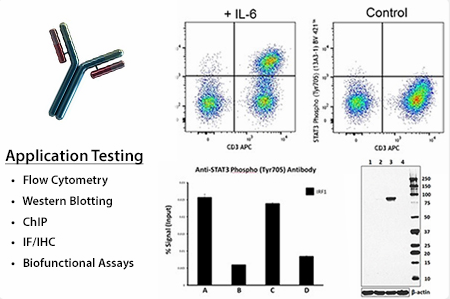
Antibody clones are then tested in a variety of assays to see which applications they are suited for. As an example, clone 13A3-1 for phosphorylated STAT3 (Tyr705) demonstrated excellent performance in flow cytometry, western blot, and chromatin immunoprecipitation.
Thus, the clone cross-validates itself by demonstrating functionality across orthogonal testing methods. Additionally, the biological induction of the phosphorylated state using IL-6 further validates the specificity of the antibody.
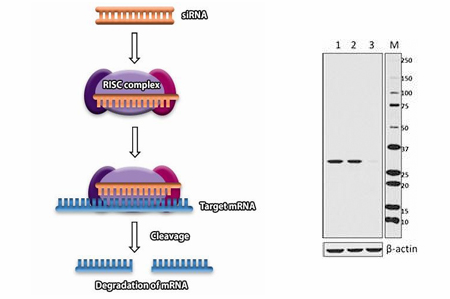
Gene Knockdown Testing
Knockout or knockdown of gene expression, such as with siRNA, is also an excellent tool for target validation. Our SIRT5 antibody, clone O91G9, was verified by western blot using HeLa cells treated with SIRT5 targeting siRNA. Lane 1 indicates untreated HeLa cells, lane 2 contains scrambled siRNA control treated cells, and lane 3 contains SIRT5-specific siRNA treated cells.
 Login / Register
Login / Register 






Follow Us