- Clone
- A17183A (See other available formats)
- Regulatory Status
- RUO
- Other Names
- Non-A beta component of AD amyloid, NACP, PARK1, Synuclein alpha-140, non-A4 component of amyloid, alpha-synuclein, isoform NACP140, non-amyloid beta component (NAP)
- Isotype
- Rat IgG2a, κ
- Ave. Rating
- Submit a Review
- Product Citations
- publications
-
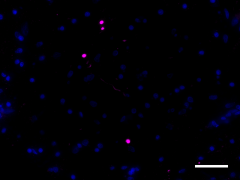
IHC staining of Biotin anti-α-Synuclein, aggregated (clone A17183A) on formalin-fixed paraffin-embedded Parkinson’s disease brain tissue. Following antigen retrieval using Sodium Citrate H.I.E.R., the tissue was incubated with 2 µg/mL of the primary antibody overnight at 4°C, followed by incubation with Alexa Fluor® 647 streptavidin (Cat No. 405237) for one hour at room temperature. Nuclei were counterstained with DAPI. The image was captured with a 40X objective. Scale bar: 50 µm -
IHC staining of Biotin anti-α-Synuclein, aggregated (clone A17183A) on formalin-fixed paraffin-embedded Parkinson’s disease brain tissue. Following antigen retrieval using Sodium Citrate H.I.E.R., the tissue was incubated with 2 µg/mL of the primary antibody overnight at 4°C, followed by incubation with Alexa Fluor® 647 streptavidin (Cat No. 405237) for one hour at room temperature. Nuclei were counterstained with DAPI. The image was captured with a 40X objective. Scale bar: 50 µm
| Cat # | Size | Price | Quantity Check Availability | Save | ||
|---|---|---|---|---|---|---|
| 864905 | 25 µg | 90€ | ||||
| 864906 | 100 µg | 212€ | ||||
α-synuclein is expressed principally in the nervous system, but it is also produced in the other tissues, including the skin. In the brain, the protein is primarily neuronal, but it is also present in glia. Neuronal α-synuclein is concentrated in the presynaptic nerve terminals, interacts with plasma membrane phospholipids, and is also present in the nuclei and mitochondria. At least three isoforms of synuclein are produced through alternative splicing. The most common isoform is a 140 amino acid-long transcript. Other isoforms are α-synuclein-126, lacking residues 41-54; and α-synuclein-112, which lacks residues 103-130. α-synuclein’s physiological role is poorly understood, but the protein has been implicated in regulating dopamine release and transport, synaptic vesicle clustering, and functioning as a SNARE-complex chaperone. α-Synuclein fibrils are a major component of the intracellular Lewy bodies that are associated with Parkinson's disease, Lewy body dementia, and multiple system atrophy.
Product DetailsProduct Details
- Verified Reactivity
- Human
- Antibody Type
- Monoclonal
- Host Species
- Rat
- Immunogen
- Recombinant human alpha-synuclein aggregates
- Formulation
- Phosphate-buffered solution, pH 7.2, containing 0.09% sodium azide
- Preparation
- The antibody was purified by affinity chromatography and conjugated with biotin under optimal conditions.
- Concentration
- 0.5 mg/mL
- Storage & Handling
- The antibody solution should be stored undiluted between 2°C and 8°C. Do not freeze.
- Application
-
IHC-P - Quality tested
- Recommended Usage
-
Each lot of this antibody is quality control tested by formalin-fixed paraffin-embedded immunohistochemical staining. For immunohistochemistry, a concentration range of 2 - 10 µg/mL is suggested. It is recommended that the reagent be titrated for optimal performance for each application.
- RRID
-
AB_2832868 (BioLegend Cat. No. 864905)
AB_2832869 (BioLegend Cat. No. 864906)
Antigen Details
- Structure
- Human α-synuclein is a 140 amino acid protein with a molecular mass of 14 kD.
- Distribution
-
Tissue distribution: Primarily nervous system, but lower expression in other tissues such as blood and skin.
Cellular distribution: Cytoskeleton, cytosol, lysosome, mitochondria, nucleus, plasma membrane, and extracellular. - Function
- α-synuclein plays several roles in synaptic activity such as synaptic vesicle trafficking and neurotransmitter release. It is also involved in the regulation of gene expression.
- Interaction
- Interacts with various proteins, such as UCHL1, phospholipase D,histones and SNARE components.
- Cell Type
- Neurons
- Biology Area
- Cell Biology, Mitochondrial Function, Neurodegeneration, Neuroinflammation, Neuroscience, Protein Misfolding and Aggregation, Protein Trafficking and Clearance, Synaptic Biology
- Molecular Family
- α-Synuclein, Presynaptic proteins, Synaptic Vesicle Trafficking/Endocytosis
- Gene ID
- 6622 View all products for this Gene ID
- UniProt
- View information about alpha-Synuclein aggregated on UniProt.org
Related FAQs
- How many biotin molecules are per antibody structure?
- We don't routinely measure the number of biotins with our antibody products but the number of biotin molecules range from 3-6 molecules per antibody.
Other Formats
View All α-Synuclein, aggregated Reagents Request Custom Conjugation| Description | Clone | Applications |
|---|---|---|
| Purified anti-α-Synuclein, aggregated | A17183A | IHC-P |
| Biotin anti-α-Synuclein, aggregated | A17183A | IHC-P |
| Alexa Fluor® 594 anti-α-Synuclein, aggregated | A17183A | IHC-P |
| Alexa Fluor® 488 anti-α-Synuclein, aggregated | A17183A | IHC-P |
Compare Data Across All Formats
This data display is provided for general comparisons between formats.
Your actual data may vary due to variations in samples, target cells, instruments and their settings, staining conditions, and other factors.
If you need assistance with selecting the best format contact our expert technical support team.
-
Purified anti-α-Synuclein, aggregated
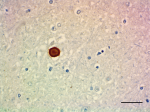
IHC staining of purified anti-α-Synuclein, aggregated antibo... 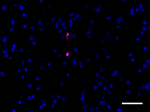
IHC staining of purified anti-α-Synuclein, aggregated antibo... 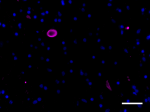
IHC staining of purified anti-α-Synuclein, aggregated antibo... -
Biotin anti-α-Synuclein, aggregated
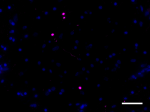
IHC staining of Biotin anti-α-Synuclein, aggregated (clone A... 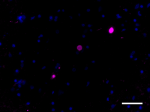
IHC staining of Biotin anti-α-Synuclein, aggregated (clone A... -
Alexa Fluor® 594 anti-α-Synuclein, aggregated
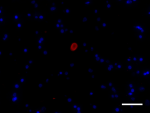
IHC staining of Alexa Fluor® 594 anti-α-Synuclein, aggregate... 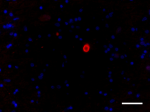
IHC staining of Alexa Fluor® 594 anti-α-Synuclein, aggregate... -
Alexa Fluor® 488 anti-α-Synuclein, aggregated

IHC staining of Alexa Fluor® 488 anti-α-Synuclein, aggregate...
 Login / Register
Login / Register 









Follow Us