- Clone
- 11B11 (See other available formats)
- Regulatory Status
- RUO
- Other Names
- Interleukin-4, Ia inducing factor (IaIF), B cell stimulating factor-1 (BSF-1), Hodgkin's cell growth factor (HCGF), Mast cell growth factor-2 (MCGF-2), Macrophage fusion factor (MFF), T cell growth factor-2 (TCGF-2)
- Isotype
- Rat IgG1, κ
- Ave. Rating
- Submit a Review
- Product Citations
- publications
Select size of product is eligible for a 40% discount! Promotion valid until September 30, 2024. Exclusions apply. To view full promotion terms and conditions or to contact your local BioLegend representative to receive a quote, visit our webpage.
IL-4 is a pleiotropic cytokine produced by activated T cells, mast cells, and basophils. IL-4 is a potent lymphoid cell growth factor which stimulates the growth and activation of certain B cells and T cells. IL-4 is important for regulation of T helper subset development.
Product DetailsProduct Details
- Verified Reactivity
- Mouse
- Antibody Type
- Monoclonal
- Host Species
- Rat
- Immunogen
- Partially purified native mouse IL-4
- Formulation
- 0.2 µm filtered in phosphate-buffered solution, pH 7.2, containing no preservative.
- Endotoxin Level
- Less than 0.01 EU/µg of the protein (< 0.001 ng/µg of the protein) as determined by the LAL test.
- Preparation
- The Ultra-LEAF™ (Low Endotoxin, Azide-Free) antibody was purified by affinity chromatography.
- Concentration
- The antibody is bottled at the concentration indicated on the vial, typically between 2 mg/mL and 3 mg/mL. Older lots may have also been bottled at 1 mg/mL. To obtain lot-specific concentration and expiration, please enter the lot number in our Certificate of Analysis online tool.
- Storage & Handling
- The antibody solution should be stored undiluted between 2°C and 8°C. This Ultra-LEAF™ solution contains no preservative; handle under aseptic conditions.
- Application
-
ELISA Capture - Quality tested
CyTOF® - Verified
ELISPOT Capture, Neut, ICC, IP, IHC - Reported in the literature, not verified in house - Recommended Usage
-
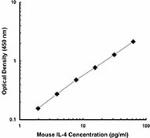
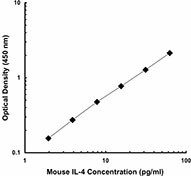
Each lot of this antibody is quality control tested by ELISA assay. For ELISPOT applications, a concentration range of 2-6 μg/ml is recommended. For ELISA capture applications, a concentration range of 0.5-2.0 μg/ml is recommended. To obtain a linear standard curve, serial dilutions of IL-4 recombinant protein ranging from 250 to 2 pg/ml are recommended for each ELISA plate. It is recommended that the reagent be titrated for optimal performance for each application.
- Application Notes
-
ELISA1,2,10,13 or ELISPOT5 Capture: The purified 11B11 antibody is useful as the capture antibody in a sandwich ELISA or ELISPOT assay, when used in conjunction with the biotinylated BVD6-24G2 antibody (Cat. No. 504202) as the detecting antibody and recombinant mouse IL-4 (Cat. No. 575609) as the standard. The LEAF™ purified antibody is suggested for ELISPOT capture.
Neutralization1-2,9,12: The 11B11 antibody can neutralize the bioactivity of natural or recombinant IL-4. The LEAF™ purified antibody (Endotoxin <0.1 EU/µg, Azide-Free, 0.2 µm filtered) is recommended for neutralization of mouse IL-4 bioactivity in vivo and in vitro (Cat. No. 504108).
Additional reported applications (for the relevant formats) include: immunoprecipitation16, immunohistochemical staining of formalin-fixed paraffin-embedded tissue sections8 and paraformaldehyde-fixed, saponin-treated frozen tissue sections6,7, and immunocytochemistry4.
Note: For testing mouse IL-4 in serum, plasma or supernatant, BioLegend's ELISA Max™ Sets (Cat. No. 431101 to 431106) are specially developed and recommended. - Additional Product Notes
-
Get a 50% discount on this product when purchased in our Activation Bundles. Restrictions apply. Learn more…
- Application References
-
- Shirai A, et al. 1994. Cytokine 6:329. (ELISA, Neut)
- Abrams J. 1995. Curr. Prot. Immunol. John Wiley and Sons New York. Unit 6.20. (ELISA, Neut)
- Assenmacher M, et al. 1994. Eur. J. Immunol. 24:1097.
- Openshaw P, et al. 1995. J. Exp. Med. 182:1357. (ICC)
- Klinman D, et al. 1994. Curr. Prot. Immunol. John Wiley and Sons New York. Unit 6.19. (ELISA Capture)
- Litton M, et al. 1994. J. Immunol. Methods 175:47. (IHC)
- Andersson U, et al. 1999. Detection and quantification of gene expression. New York:Springer-Verlag. (IHC)
- Fan WY, et al. 2001. Exp. Biol. Med. 226:1045. (IHC)
- Hara M, et al. 2001. J. Immunol. 166:3789. (Neut)
- Dzhagalov I, et al. 2007. J. Immunol. 178:2113. (ELISA)
- Lawson BR, et al. 2007. J. Immunol. 178:5366.
- Wang W, et al. 2007. J. Immunol. 178:4885. (Neut)
- Xu G, et al. 2007. J. Immunol. 179:5358. (ELISA) PubMed
- Ohnmacht C, et al. 2008. Blood 113:2816. PubMed
- Charles N, et al. 2010. Nat. Med. 16:701. (FC) PubMed
- Zavorotinskaya T, et al. 2003. Mol. Ther. 7:155. (IP)
- Product Citations
-
- RRID
-
AB_11149679 (BioLegend Cat. No. 504121)
AB_11150601 (BioLegend Cat. No. 504122)
AB_2750404 (BioLegend Cat. No. 504135)
AB_2750405 (BioLegend Cat. No. 504136)
AB_2750406 (BioLegend Cat. No. 504137)
AB_2750407 (BioLegend Cat. No. 504138)
Antigen Details
- Structure
- Cytokine; 15-19 kD (Mammalian)
- Bioactivity
- Differentiation of naïve CD4+ T cells to the TH2 type, proliferation/differentiation of activated B cells, expression of class II MHC antigens, and of low affinity IgE receptors in resting B cells
- Cell Sources
- Mast cells, T cells, bone marrow stromal cells
- Cell Targets
- B cells, T cells, monocytes, endothelial cells, fibroblasts
- Receptors
- Heterodimer IL-4Rα (CD124); γ-subunit (CD132) in common with IL-2R, IL-7R, IL-13R, IL-15R
- Cell Type
- Tregs
- Biology Area
- Immunology
- Molecular Family
- Cytokines/Chemokines
- Antigen References
-
1. Fitzgerald K, et al. Eds. 2001. The Cytokine FactsBook. Academic Press San Diego.
2. Boulay J, et al. 1992. Curr. Opin. Immunol. 4:294.
3. Dullens H, et al. 1991. In vivo 5:567.
4. Paul W. 1991. Blood 77:1859. - Regulation
- Upregulated by IL-2, platelet activating factor; downregulated by TGF-β
- Gene ID
- 16189 View all products for this Gene ID
- UniProt
- View information about IL-4 on UniProt.org
Related FAQs
- Do you guarantee that your antibodies are totally pathogen free?
-
BioLegend does not test for pathogens in-house aside from the GoInVivo™ product line. However, upon request, this can be tested on a custom basis with an outside, independent laboratory.
- Does BioLegend test each Ultra-LEAF™ antibody by functional assay?
-
No, BioLegend does not test Ultra-LEAF™ antibodies by functional assays unless otherwise indicated. Due to the possible complexities and variations of uses of biofunctional antibodies in different assays and because of the large product portfolio, BioLegend does not currently perform functional assays as a routine QC for the antibodies. However, we do provide references in which the antibodies were used for functional assays and we do perform QC to verify the specificity and quality of the antibody based on our strict specification criteria.
- Does BioLegend test each Ultra-LEAF™ antibody for potential pathogens?
-
No, BioLegend does not test for pathogens in-house unless otherwise indicated. However, we can recommend an outside vendor to perform this testing as needed.
- Have you tested this Ultra-LEAF™ antibody for in vivo or in vitro applications?
-
We don't test our antibodies for in vivo or in vitro applications unless otherwise indicated. Depending on the product, the TDS may describe literature supporting usage of a particular product for bioassay. It may be best to further consult the literature to find clone specific information.
Other Formats
View All IL-4 Reagents Request Custom Conjugation| Description | Clone | Applications |
|---|---|---|
| APC anti-mouse IL-4 | 11B11 | ICFC |
| PE anti-mouse IL-4 | 11B11 | ICFC |
| Purified anti-mouse IL-4 | 11B11 | ELISA Capture,CyTOF®,IP,IHC,ICC |
| Alexa Fluor® 488 anti-mouse IL-4 | 11B11 | ICFC |
| Alexa Fluor® 647 anti-mouse IL-4 | 11B11 | ICFC |
| PE/Cyanine7 anti-mouse IL-4 | 11B11 | ICFC |
| Brilliant Violet 421™ anti-mouse IL-4 | 11B11 | ICFC |
| Ultra-LEAF™ Purified anti-mouse IL-4 | 11B11 | ELISA Capture,CyTOF®,ELISPOT Capture,Neut,ICC,IP,IHC |
| PerCP/Cyanine5.5 anti-mouse IL-4 | 11B11 | ICFC |
| Brilliant Violet 605™ anti-mouse IL-4 | 11B11 | ICFC |
| Purified anti-mouse IL-4 (Maxpar® Ready) | 11B11 | ELISA Capture,CyTOF® |
| PE/Dazzle™ 594 anti-mouse IL-4 | 11B11 | ICFC |
| Brilliant Violet 711™ anti-mouse IL-4 | 11B11 | ICFC |
| APC/Fire™ 750 anti-mouse IL-4 | 11B11 | ICFC |
Customers Also Purchased
Compare Data Across All Formats
This data display is provided for general comparisons between formats.
Your actual data may vary due to variations in samples, target cells, instruments and their settings, staining conditions, and other factors.
If you need assistance with selecting the best format contact our expert technical support team.
-
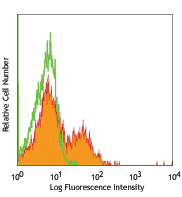
APC anti-mouse IL-4

PMA+ionomycin-stimulated (6 hours, in presence of brefeldin ... 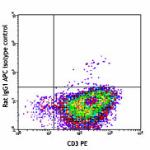
-
PE anti-mouse IL-4
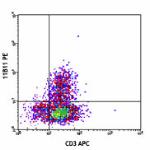
PMA+ionomycin-stimulated (6 hours, in presence of brefeldin ... 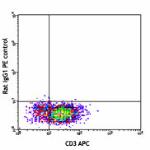
-
Purified anti-mouse IL-4
-
Alexa Fluor® 488 anti-mouse IL-4

PMA+ionomycin-stimulated (6 hours, in presence of brefeldin ... 
-
Alexa Fluor® 647 anti-mouse IL-4

PMA+ionomycin-stimulated (6 hours, in presence of brefeldin ... 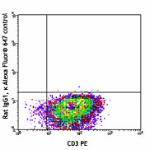
-
PE/Cyanine7 anti-mouse IL-4

PMA+ionomycin-stimulated (in presence of Brefeldin A for 6 h... -
Brilliant Violet 421™ anti-mouse IL-4

PMA+ionomycin-stimulated (6 hours, in presence of brefeldin ... 
-
Ultra-LEAF™ Purified anti-mouse IL-4

-
PerCP/Cyanine5.5 anti-mouse IL-4

Th2-polarized cells from C57BL/6 mouse T cells were stimulat... -
Brilliant Violet 605™ anti-mouse IL-4

PMA+ionomycin-stimulated (6 hours, in presence of brefeldin ... 
-
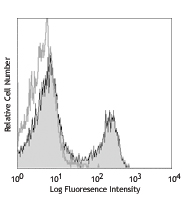
Purified anti-mouse IL-4 (Maxpar® Ready)

C57BL/6 mouse splenocytes were incubated for 20 hours in med... 
-
PE/Dazzle™ 594 anti-mouse IL-4

PMA+ionomycin-stimulated (6 hours, in the presence of brefel... 
-
Brilliant Violet 711™ anti-mouse IL-4

PMA + ionomycin-stimulated (six hours, in presence of brefel... 
-
APC/Fire™ 750 anti-mouse IL-4

PMA+ionomycin-stimulated (6 hours, in presence of brefeldin ...
 Login / Register
Login / Register 











Follow Us