- Regulatory Status
- RUO
- Other Names
- Interleukin 7, Lymphopoietin-1 (LP-1)
- Ave. Rating
- Submit a Review
- Product Citations
- publications
-
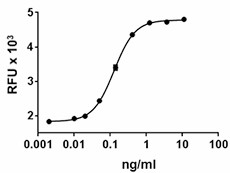
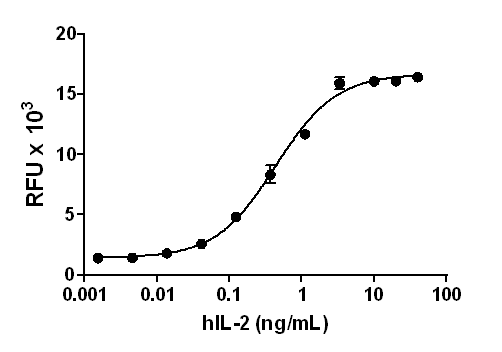
Proliferation of PHA-activated human PBL induced by human IL-7.
IL-7 was initially described as a stromal derived factor which is capable of inducing the growth of pre-B cells in vitro. IL-7 acts on a variety of cells through its receptor (IL-7R), a heterodimer consisting of IL-7Rα (CD127) and a common γc chain (CD132) shared by other cytokine (IL-2, IL-4, IL-9, IL-15, and IL-21) receptors. In addition, IL-7Rα is shared with TSLP. The generation of IL-7-deficient and IL-7Rα-deficient mice and monoclonal antibody blocking experiments confirmed the requirement of IL-7 for B-cell development in mice. Nevertheless, mutations in the α chain of the IL-7 receptor in patients with severe combined immunodeficiency (SCID) confirmed that IL-7 is indispensable for T-cell development in humans. However, the presence of B cells in these individuals suggests important differences between the role of IL-7 in murine and human lymphocyte development. Thus, although human B-cell development does not appear to require IL-7, immature human B cells do proliferate in response to IL-7. Nevertheless, most recent information suggests that IL-7 dependence in human lymphopoiesis increases during the progression of ontogeny in cord blood and bone marrow. IL-7 can be associated to hepatocyte grow factor (HGFβ) to form a hybrid cytokine (IL-7/HGFβ), which induces greater proliferation of CFU-S, SLPs, and pre–pro-B cells than does native IL-7. The hybrid cytokine signals through both IL-7R (IL-7Rα plus γc) and c-Met. IL-7 has a thymic antiapoptotic effect and induces the expression of antiapoptotic proteins Bcl-2 and Mcl-1 and the inhibition of proapoptotic proteins Bax and Bad. In addition, IL-7 is a key regulator of glucose uptake in T lymphocytes. TGF-β has been shown to down-regulate IL-7 mRNA and protein secretion from human bone marrow stromal cells. In addition, TGF-β inhibits IL-7-induced proliferation of pre-B cells.
Product DetailsProduct Details
- Source
- Human IL-7, amino acids Asp26-His177 (Accession# NP_000871.1) was expressed in 293E cells.
- Molecular Mass
- The 165 amino acid recombinant protein has a predicted molecular mass of approximately 18.8 kD. The DTT-reduced and non-reduced protein migrate at approximately 20- 30 kD by SDS-PAGE. The C-terminal contains an 8His-(TG8HGGQ)-tag.
- Purity
- > 95%, as determined by Coomassie stained SDS-PAGE.
- Formulation
- 0.22 µm filtered protein solution is in PBS.
- Endotoxin Level
- Less than 0.01 ng per µg cytokine as determined by the LAL method.
- Concentration
- 10 and 25 µg sizes are bottled at 200 µg/mL. 100 µg size and larger sizes are lot-specific and bottled at the concentration indicated on the vial. To obtain lot-specific concentration and expiration, please enter the lot number in our Certificate of Analysis online tool.
- Storage & Handling
- Unopened vial can be stored between 2°C and 8°C for up to 2 weeks, at -20°C for up to six months, or at -70°C or colder until the expiration date. For maximum results, quick spin vial prior to opening. The protein can be aliquoted and stored at -20°C or colder. Stock solutions can also be prepared at 50 - 100 µg/mL in appropriate sterile buffer, carrier protein such as 0.2 - 1% BSA or HSA can be added when preparing the stock solution. Aliquots can be stored between 2°C and 8°C for up to one week and stored at -20°C or colder for up to 3 months. Avoid repeated freeze/thaw cycles.
- Activity
- ED50 =0.2 — 1.2 ng/ml, corresponding to a specific activity of 0.83 - 5 x 106 units/mg, as determined by PHA-activated human PBL proliferation induced by human IL-7 in a dose dependent manner.
- Application
-
Bioassay
- Application Notes
-
BioLegend carrier-free recombinant proteins provided in liquid format are shipped on blue-ice. Our comparison testing data indicates that when handled and stored as recommended, the liquid format has equal or better stability and shelf-life compared to commercially available lyophilized proteins after reconstitution. Our liquid proteins are verified in-house to maintain activity after shipping on blue ice and are backed by our 100% satisfaction guarantee. If you have any concerns, contact us at tech@biolegend.com.
- Product Citations
-
Antigen Details
- Distribution
-
IL-7 is produced by epithelial cells in thymus, bone marrow, and intestine. Additional sites of IL-7 production include epithelial goblet cells, keratinocytes, fetal liver, adult liver, dendritic cells, skeletal muscle cells, fibroblastic reticular cells, and follicular dendritic cells.
- Function
- IL-7 induces proliferation of human immature B cells, and it is critical for T-cell development and peripheral T-cell homeostasis.
- Interaction
- IL-7Rα is expressed on immature B cells through the early pre-B stage. The IL-77Rα is widely expressed in T cells, CD4, CD8 naïve and memory cells, and dendritic cells.
- Ligand/Receptor
- IL-7Rα (CD127), common gamma chain (γc or CD132).
- Cell Type
- B cells, Hematopoietic stem and progenitors
- Biology Area
- Cell Biology, Immunology, Stem Cells
- Molecular Family
- Cytokines/Chemokines
- Antigen References
-
1. Park LS, et al. 1990. J. Exp. Med. 171:1073.
2. Fry TJ and Mackall CL, 2002. Blood 99:3892.
3. Lai L, et al. 2006. Blood 107:1776.
4. Link A, et al. 2007. Nat. Immunol. 8:1255.
5. Wofford J, et al. 2008. Blood 111:2101.
6. Parrish YK, et al. 2009. J. Immunol. 182:4255.
7. Saini M, et al. 2009. Blood 113:5793.
8. Guimond M, et al. 2009. Nat. Immunol. 10:149. - Gene ID
- 3574 View all products for this Gene ID
- UniProt
- View information about IL-7 on UniProt.org
Related FAQs
- Why choose BioLegend recombinant proteins?
-
• Each lot of product is quality-tested for bioactivity as indicated on the data sheet.
• Greater than 95% Purity or higher, tested on every lot of product.
• 100% Satisfaction Guarantee for quality performance, stability, and consistency.
• Ready-to-use liquid format saves time and reduces challenges associated with reconstitution.
• Bulk and customization available. Contact us.
• Learn more about our Recombinant Proteins. - How does the activity of your recombinant proteins compare to competitors?
-
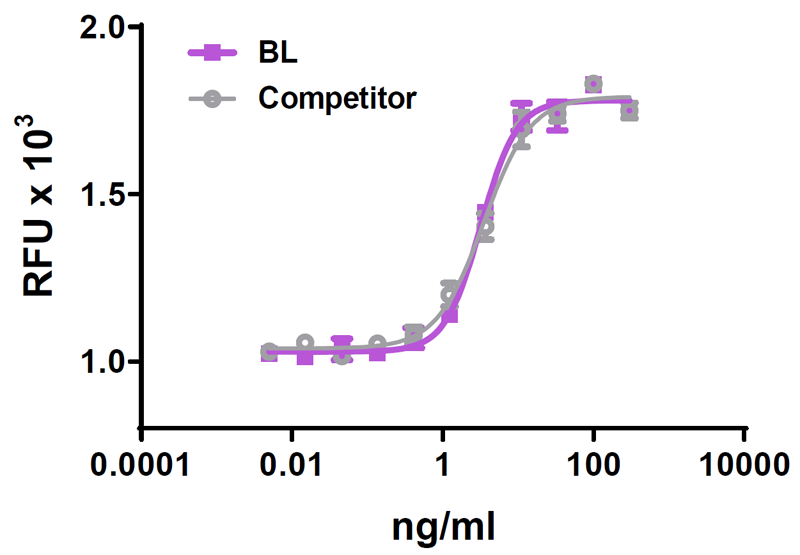
We quality control each and every lot of recombinant protein. Not only do we check its bioactivity, but we also compare it against other commercially available recombinant proteins. We make sure each recombinant protein’s activity is at least as good as or better than the competition’s. In order to provide you with the best possible product, we ensure that our testing process is rigorous and thorough. If you’re curious and eager to make the switch to BioLegend recombinants, contact your sales representative today!
- What is the specific activity or ED50 of my recombinant protein?
-
The specific activity range of the protein is indicated on the product datasheets. Because the exact activity values on a per unit basis can largely fluctuate depending on a number of factors, including the nature of the assay, cell density, age of cells/passage number, culture media used, and end user technique, the specific activity is best defined as a range and we guarantee the specific activity of all our lots will be within the range indicated on the datasheet. Please note this only applies to recombinants labeled for use in bioassays. ELISA standard recombinant proteins are not recommended for bioassay usage as they are not tested for these applications.
- Have your recombinants been tested for stability?
-
Our testing shows that the recombinant proteins are able to withstand room temperature for a week without losing activity. In addition the recombinant proteins were also found to withstand four cycles of freeze and thaw without losing activity.
- Does specific activity of a recombinant protein vary between lots?
-
Specific activity will vary for each lot and for the type of experiment that is done to validate it, but all passed lots will have activity within the established ED50 range for the product and we guarantee that our products will have lot-to-lot consistency. Please conduct an experiment-specific validation to find the optimal ED50 for your system.
- How do you convert activity as an ED50 in ng/ml to a specific activity in Units/mg?
-
Use formula Specific activity (Units/mg) = 10^6/ ED50 (ng/mL)
 Login / Register
Login / Register 









Follow Us