- Clone
- BVD2-21C11 (See other available formats)
- Regulatory Status
- RUO
- Other Names
- Granulocyte/macrophage colony-stimulating factor, CSF-α, Pluripoietin-α, Eosinophil colony stimulating factor (Eo-CSF), Burst promoting activity (BPA)
- Isotype
- Rat IgG2a, κ
- Ave. Rating
- Submit a Review
- Product Citations
- publications
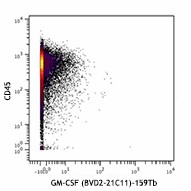
-

Human PBMCs were incubated for 6 hours in media alone (top) or with PMA and Ionomycin (bottom) in the presence of monensin and brefeldin A. Cells were then fixed, permeabilized, and stained with 154Sm-anti-CD45 (HI30) and 159Tb-anti-GM-CSF (BVD2-21C11). Data provided by DVS Sciences. -

-

| Cat # | Size | Price | Quantity Check Availability | Save | ||
|---|---|---|---|---|---|---|
| 502315 | 100 µg | $171 | ||||
Granulocyte/macrophage - colony stimulating factor (GM-CSF) is a hematopoietic factor that is produced by activated T cells, B cells, mast cells, macrophages, fibroblasts, and endothelial cells. In addition to supporting colony formation of granulocyte/macrophage progenitors, GM-CSF is a growth factor for erythroid, megakaryocyte, and eosinophil progenitors.
Product DetailsProduct Details
- Verified Reactivity
- Human
- Reported Reactivity
- Cynomolgus, Rhesus
- Antibody Type
- Monoclonal
- Host Species
- Rat
- Immunogen
- E. coli-expressed, recombinant human GM-CSF.
- Formulation
- Phosphate-buffered solution, pH 7.2, containing 0.09% sodium azide and EDTA.
- Preparation
- The antibody was purified by affinity chromatography.
- Concentration
- 1.0 mg/ml
- Storage & Handling
- The antibody solution should be stored undiluted between 2°C and 8°C.
- Application
-
ELISA Capture - Quality tested
CyTOF® - Verified - Recommended Usage
-
This product is suitable for use with the Maxpar® Metal Labeling Kits. For metal labeling using Maxpar® Ready antibodies, proceed directly to the step to Partially Reduce the Antibody by adding 100 µl of Maxpar® Ready antibody to 100 µl of 4 mM TCEP-R in a 50 kDa filter and continue with the protocol. Always refer to the latest version of Maxpar® User Guide when conjugating Maxpar® Ready antibodies.
- Application Notes
-
Additional reported applications (for the relevant formats) include: ELISA1-4 or ELISPOT3,4 Detection, Neutralization1, immunohistochemical staining of paraformaldehyde-fixed, saponin-treated5,6 and acetone-fixed7 frozen tissue sections, and immunocytochemistry.
Note: For testing human GM-CSF in serum or plasma, BioLegend's ELISA Max™ Sets (Cat. No. 432001 to 432006) are specially developed and recommended.
- Additional Product Notes
-
Maxpar® is a registered trademark of Standard BioTools Inc.
-
Application References
(PubMed link indicates BioLegend citation) -
- Abrams J, et al. 1992. Immunol. Rev. 127:5. (ELISA, Neut, IP)
- Abrams J, et al. 1994. Eosinophils in Allergy and Inflammation. Marcel Dekker New York. p.133. (ELISA)
- Bacchetta R, et al. 1990. J. Immunol. 144:902. (ELISA)
- Kita H, et al. 1991. J. Exp. Med. 174:745. (ELISA)
- Andersson U, et al. 1999. Detection and quantification of gene expression. New York:Springer-Verlag. (IHC)
- Andersson J, et al. 1994. Immunology 83:16. (IHC)
- Rasouli J, et al. 2015. J. Immunol. 11:5085-93. (IHC)
- RRID
-
AB_2563770 (BioLegend Cat. No. 502315)
Antigen Details
- Structure
- Cytokine; 22 kD (Mammalian)
- Bioactivity
- Growth/development factor for granulocyte/macrophage progenitors; differentiates myeloblasts/monoblasts; synergizes with Epo proliferation of erythroid/megakaryocytic progenitors
- Cell Sources
- T cells, macrophages, fibroblasts, endothelial cells, mast cells
- Cell Targets
- Granulocyte/macrophage/erythroid/megakaryocytic progenitors, myeloblasts, monoblasts
- Receptors
- Heterodimer GM-CSFR α subunit (CDw116); β-subunit (CDw131), which is also shared with the IL-3 and IL-5 receptor α chains
- Biology Area
- Cell Biology, Stem Cells
- Molecular Family
- Cytokines/Chemokines, Growth Factors
- Antigen References
-
1. Fitzgerald K, et al. Eds. 2001. The Cytokine FactsBook. Academic Press San Diego.
2. Demetri G, et al. 1991. Blood 78:2791.
3. Fan D, et al. 1991. In vivo 5:571.
4. Negrin R, et al. 1992. Adv. Pharmacol. 23:263. - Regulation
- Synergistic with IL-1, IL-3, G-CSF; E21R competitive antagonist for receptor binding; stored in ECM with heparan sulfate proteoglycans
- Gene ID
- 1437 View all products for this Gene ID
- UniProt
- View information about GM-CSF on UniProt.org
Related FAQs
- Can I obtain CyTOF data related to your Maxpar® Ready antibody clones?
-
We do not test our antibodies by mass cytometry or on a CyTOF machine in-house. The data displayed on our website is provided by Fluidigm®. Please contact Fluidigm® directly for additional data and further details.
- Can I use Maxpar® Ready format clones for flow cytometry staining?
-
We have not tested the Maxpar® Ready antibodies formulated in solution containing EDTA for flow cytometry staining. While it is likely that this will work in majority of the situations, it is best to use the non-EDTA formulated version of the same clone for flow cytometry testing. The presence of EDTA in some situations might negatively affect staining.
- I am having difficulty observing a signal after conjugating a metal tag to your Maxpar® antibody. Please help troubleshoot.
-
We only supply the antibody and not test that in house. Please contact Fluidigm® directly for troubleshooting advice: http://techsupport.fluidigm.com/
- Is there a difference between buffer formulations related to Maxpar® Ready and purified format antibodies?
-
The Maxpar® Ready format antibody clones are formulated in Phosphate-buffered solution, pH 7.2, containing 0.09% sodium azide and EDTA. The regular purified format clones are formulated in solution that does not contain any EDTA. Both formulations are however without any extra carrier proteins.
Other Formats
View All GM-CSF Reagents Request Custom Conjugation| Description | Clone | Applications |
|---|---|---|
| Biotin anti-human GM-CSF | BVD2-21C11 | ELISA Detection,ELISPOT Detection,ICFC |
| PE anti-human GM-CSF | BVD2-21C11 | ICFC |
| Purified anti-human GM-CSF | BVD2-21C11 | ELISA Capture,ICFC,IP,WB |
| PerCP/Cyanine5.5 anti-human GM-CSF | BVD2-21C11 | ICFC |
| APC anti-human GM-CSF | BVD2-21C11 | ICFC |
| Pacific Blue™ anti-human GM-CSF | BVD2-21C11 | ICFC |
| Purified anti-human GM-CSF (Maxpar® Ready) | BVD2-21C11 | ELISA Capture,CyTOF® |
| PE/Dazzle™ 594 anti-human GM-CSF | BVD2-21C11 | ICFC |
| Ultra-LEAF™ Purified anti-human GM-CSF | BVD2-21C11 | Neut |
| TotalSeq™-B0943 anti-human GM-CSF | BVD2-21C11 | ICPG |
| Brilliant Violet 421™ anti-human GM-CSF | BVD2-21C11 | ICFC |
Compare Data Across All Formats
This data display is provided for general comparisons between formats.
Your actual data may vary due to variations in samples, target cells, instruments and their settings, staining conditions, and other factors.
If you need assistance with selecting the best format contact our expert technical support team.
-
Biotin anti-human GM-CSF
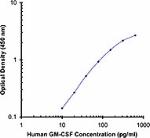
-
PE anti-human GM-CSF
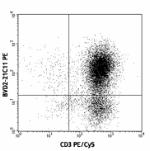
PMA and ionomycin-stimulated Th2 polarized human peripheral ... -
Purified anti-human GM-CSF
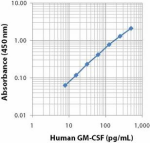
BVD2-21C11_PURE_GM-CSF_ELISA_092117 -
PerCP/Cyanine5.5 anti-human GM-CSF

PMA and ionomycin-stimulated Th2-polarized human peripheral ... -
APC anti-human GM-CSF
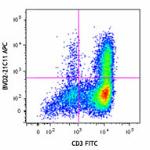

-
Pacific Blue™ anti-human GM-CSF

PMA and ionomycin-stimulated Th2-polarized human peripheral ... 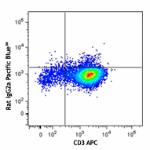
-
Purified anti-human GM-CSF (Maxpar® Ready)
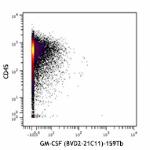
Human PBMCs were incubated for 6 hours in media alone (top) ... 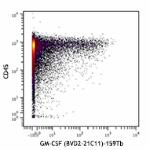

-
PE/Dazzle™ 594 anti-human GM-CSF

PMA and ionomycin-stimulated Th2-polarized human peripheral ... 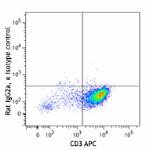
-
Ultra-LEAF™ Purified anti-human GM-CSF

Recombinant human GM-CSF (Black Circles, Cat. No. 572904) i... -
TotalSeq™-B0943 anti-human GM-CSF
-
Brilliant Violet 421™ anti-human GM-CSF

PMA+ ionomycin stimulated (6 hours) human peripheral blood l...
 Login/Register
Login/Register 













Follow Us