- Clone
- A20098E (See other available formats)
- Regulatory Status
- RUO
- Other Names
- LPS-induced CXC chemokine (LIX), granulocyte chemotactic protein-2 (GCP-2), alveolar macrophage chemotactic factor-II (AMCF-II), epithelial neutrophil-activating protein (ENA78)
- Isotype
- Rat IgG2b, λ
- Ave. Rating
- Submit a Review
- Product Citations
- publications
-

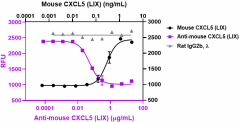
Recombinant mouse CXCL5 (LIX) (Cat. No. 573302) (black circles) induces the chemotaxis of mouse Baf3-mCXCR2 transfectant chemoattractant cells. Ultra-LEAF™ purified anti-mouse CXCL5 (LIX) (clone A20098E) (purple squares) neutralizes recombinant mouse CXCL5 (LIX)-induced chemotaxis at 0.8 ng/mL on mouse Baf3-mCXCR2 cells in a dose-dependent manner, whereas the LEAF™ purified rat IgG2b, λ isotype control (gray triangles) does not have the effect. ND50 range: 0.025 - 0.25 µg/mL
| Cat # | Size | Price | Quantity Check Availability | Save | ||
|---|---|---|---|---|---|---|
| 662053 | 100 µg | $358 | ||||
| 662054 | 1 mg | $1205 | ||||
CXCL5 is an ELR-CXC chemokine characterized by a Glu-Leu-Arg (ELR) motif preceding the characteristic CXC sequence. CXCL5 chemoattracts and activates neutrophils during inflammation through the binding to CXCR2. CXCL5 exists in different NH2- and COOH-truncated forms, and both NH2-terminal and COOH-terminal truncation result in an increase in neutrophil chemotactic potency. CXCL5 (8–78) and CXCL5 (9–78) induce a higher neutrophil influx in vivo compared with CXCL5 (1–78). CXCL5 regulates the availability of binding sites for other ELR-CXC chemokines released during inflammation, through its interaction with erythrocyte duffy antigen receptor (DARC). As a result, CXCL5 induces the increase of CXCL1 and CXCL2 in a model of pulmonary inflammation. Therefore, CXCL5 acts as a regulator of chemokine scavenging and pulmonary host defense to bacterial infection. Lung epithelial cells express CXCR2. Consequently, CXCL5 may modulate neutrophil transepithelial and transendothelial migration. Mouse CXCL5 is the ortholog of human CXCL6.
Product DetailsProduct Details
- Verified Reactivity
- Mouse
- Antibody Type
- Monoclonal
- Host Species
- Rat
- Immunogen
- Recombinant mouse CXCL5 (LIX)
- Formulation
- 0.2 µm filtered in phosphate-buffered solution, pH 7.2, containing no preservative.
- Endotoxin Level
- Less than 0.01 EU/µg of the protein (< 0.001 ng/µg of the protein) as determined by the LAL test.
- Preparation
- The Ultra-LEAF™ (Low Endotoxin, Azide-Free) antibody was purified by affinity chromatography.
- Concentration
- The antibody is bottled at the concentration indicated on the vial, typically between 2 mg/mL and 3 mg/mL. To obtain lot-specific concentration and expiration, please enter the lot number in our Certificate of Analysis online tool.
- Storage & Handling
- The antibody solution should be stored undiluted between 2°C and 8°C. This Ultra-LEAF™ solution contains no preservative; handle under aseptic conditions.
- Application
-
Neut - Quality tested
- Recommended Usage
-
Each lot of this antibody is quality control tested by neutralizing the chemotaxis induced by recombinant mouse CXCL5 (LIX) (Cat. No. 573302) at 0.8 ng/mL on mouse Baf3- mCXCR2 transfectant chemoattractant cells. ND50 range: 0.025 - 0.25 µg/mL. It is recommended that the reagent be titrated for optimal performance for each application.
- RRID
-
AB_2910492 (BioLegend Cat. No. 662053)
AB_2910492 (BioLegend Cat. No. 662054)
Antigen Details
- Structure
- Chemokine
- Distribution
-
Endothelial cells, fibroblasts, monocytes, macrophages, platelets, alveolar type II cells
- Function
- CXCL5 chemoattracts and activates neutrophils during inflammation, and it is induced by LPS and inflammatory cytokines. NH2-terminal truncation enhances, whereas citrullination decreases the chemotactic properties of CXCL5.
- Interaction
- Neutrophils
- Ligand/Receptor
- CXCR2
- Antigen References
-
- Wuyts A, et al. 1999. J Immunol. 163:6155-63.
- Jeyaseelan S, et al. 2005. Am J Respir Cell Mol Biol. 32:531-9.
- Mei J, et al. 2010. Immunity. 33:106-17.
- Montier A, et al. 2010. J Biol Chem. 285:29750-9.
- Grommes J and Soehnlein O. 2011. Mol Med. 17:293-307.
- Gene ID
- 20304 View all products for this Gene ID
- UniProt
- View information about CXCL5 on UniProt.org
Related Pages & Pathways
Pages
Related FAQs
- Do you guarantee that your antibodies are totally pathogen free?
-
BioLegend does not test for pathogens in-house aside from the GoInVivo™ product line. However, upon request, this can be tested on a custom basis with an outside, independent laboratory.
- Does BioLegend test each Ultra-LEAF™ antibody by functional assay?
-
No, BioLegend does not test Ultra-LEAF™ antibodies by functional assays unless otherwise indicated. Due to the possible complexities and variations of uses of biofunctional antibodies in different assays and because of the large product portfolio, BioLegend does not currently perform functional assays as a routine QC for the antibodies. However, we do provide references in which the antibodies were used for functional assays and we do perform QC to verify the specificity and quality of the antibody based on our strict specification criteria.
- Does BioLegend test each Ultra-LEAF™ antibody for potential pathogens?
-
No, BioLegend does not test for pathogens in-house unless otherwise indicated. However, we can recommend an outside vendor to perform this testing as needed.
- Have you tested this Ultra-LEAF™ antibody for in vivo or in vitro applications?
-
We don't test our antibodies for in vivo or in vitro applications unless otherwise indicated. Depending on the product, the TDS may describe literature supporting usage of a particular product for bioassay. It may be best to further consult the literature to find clone specific information.
Other Formats
View All CXCL5 Reagents Request Custom Conjugation| Description | Clone | Applications |
|---|---|---|
| Ultra-LEAF™ Purified anti-mouse CXCL5 (LIX) | A20098E | Neut |
Compare Data Across All Formats
This data display is provided for general comparisons between formats.
Your actual data may vary due to variations in samples, target cells, instruments and their settings, staining conditions, and other factors.
If you need assistance with selecting the best format contact our expert technical support team.
-
Ultra-LEAF™ Purified anti-mouse CXCL5 (LIX)

Recombinant mouse CXCL5 (LIX) (Cat. No. 573302) (black circl...
 Login/Register
Login/Register 








Follow Us