- Regulatory Status
- RUO
- Other Names
- Beta-Secretase-1, Beta-site APP-cleaving enzyme 1, aspartyl protease 2 (ASP-2), Memapsin 2, Secretase, BACE
- Ave. Rating
- Submit a Review
- Product Citations
- publications
-

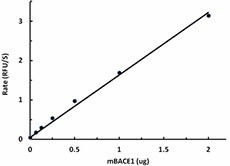
The activity of mouse BACE1 was measured with 10 µM of fluorogenic substrate, Mca-SEVNLDAEFRK(Dnp)RR-NH2 , in the presence of 0.063, 0.125, 0.25, 0.5, 1, 2 µg of mBACE1. The activity of mBACE1 is greater than 8.0 pmole/min/µg.
| Cat # | Size | Price | Quantity Check Availability | Save | ||
|---|---|---|---|---|---|---|
| 765802 | 10 µg | $112 | ||||
| 765804 | 25 µg | $176 | ||||
Beta-site APP-cleaving enzyme 1 (BACE1), also known as beta secretase 1, aspartyl protease 2 (ASP-2) and memapsin 2, is a type I transmembrane aspartic protease with 501 amino acid that is related to the pepsin family and the retroviral aspartic proteases. The BACE1 catalytic domain contains aspartic protease active site motifs at amino acids 93–96 and 289–292, with each motif containing the highly conserved sequence defining aspartic proteases, D(T/S)G(T/S). It has a typical acidic pH optimum of aspartic protease. BACE1 is expressed at low levels in most cell types of the body, although it is more highly expressed in neurons. In the brain, this protein is expressed highly in the substantia nigra, locus coruleus and medulla oblongata. It is localized within acidic intracellular compartments including endosomes and trans-Golgi network (TGN). BACE1 is responsible for the proteolytic processing of the amyloid precursor protein (APP). BACE-1 is the peptidase predominantly responsible for cleavage of the amyloid precursor protein beta site in the brain to generate the amyloid beta peptide. BACE-1 cleaves at the N-terminus of the a-beta peptide sequence, between residues 671 and 672 of APP, leads to the generation and extracellular release of beta-cleaved soluble APP, and a corresponding cell-associated C-terminal fragment which is later released by gamma-secretase. The physiological function of BACE1 is not clearly understood. A potential role for BACE1 exists in modulating sodium currents, as evidenced by cleavage of voltage-gated Na+ channel accessory subunits (Navβ1-4). It has been implicated in the regulation of myelination and myelin sheath thickness and in the control of motor coordination via cleavage of neuregulin-1 and neuregulin-3, and also in axon guidance via cleavage of close Homolog of L1 (CHL1).
Product DetailsProduct Details
- Source
- Mouse BACE1, amino acids Thr22-Thr457 (Accession #NM_011792) with a C-terminal TG-8H-GGQ tag was expressed in CHO cells.
- Molecular Mass
- The 448 amino acid recombinant protein has a predicted molecular mass of approximately 49.8 kDa. The non-reduced and DTT-reduced proteins migrate at 50 - 65 and 60 - 70 kDa, respectively, by SDS-PAGE.
- Purity
- > 95%, as determined by Coomasie stained SDS-PAGE
- Formulation
- 0.22 µm filtered protein solution is in pH 7.5 buffer containing 20 mM TRIS, 300 mM NaCl, and 1 mM EDTA.
- Endotoxin Level
- Less than 1.0 EU per µg of protein as determined by the LAL method
- Concentration
- 10-100 µg sizes are bottled at 200 µg/mL.
- Storage & Handling
- Unopened vial can be stored at -20 °C or -70°C for six months. For maximum results, quick spin vial prior to opening. Avoid repeated freeze/thaw cycles.
- Activity
- The activity of mouse BACE1 is measured with 10 µM of fluorogenic substrate, Mca-SEVNLDAEFRK(Dnp)RR-NH2 with a specific activity value greater than 8.0 pmol/µg/min.
- Application
-
Bioassay
- Application Notes
-
Mouse BACE1 Activity assay
Mouse BACE1 activity is measured by its ability to cleave a fluorogenic peptide substrate Mca-SEVNLDAEFRK(Dnp)RR-NH2. The increase of the product is monitored by increase in intensity of emission at 405 nm with excitation at 320 nm. This enzyme is in active form and does not require further activation.
Materials
- Assay Buffer: pH 4.0, 50 mM Citrate, 0.1% BSA
- Recombinant Human BACE1
- BACE1 Substrate: Mca-SEVNLDAEFRK(Dnp)RR-NH2
Activity assay procedures
- Dilute mBACE1 in the assay buffer at 10 µg/mL (10 ng/µL).
- Dilute the substrate at 10 µM in Assay Buffer.
- Load into a well plate 50 μL of the 10 ng/μL mBACE1 and start the reaction by adding 50 μL of 20 μM Substrate. Include a substrate blank containing 50 μL of Assay Buffer and 50 μL of 20 μM Substrate solution.
- Read the product formation by measuring 320/405 nm (Excitation/Emission) in kinetic mode for 5 minutes.
The final mBACE1 concentration is 5 µg/mL (0.5 µg) and the substrate is 10 µM.
BioLegend carrier-free recombinant proteins provided in liquid format are shipped on blue-ice. Our comparison testing data indicates that when handled and stored as recommended, the liquid format has equal or better stability and shelf-life compared to commercially available lyophilized proteins after reconstitution. Our liquid proteins are verified in-house to maintain activity after shipping on blue ice and are backed by our 100% satisfaction guarantee. If you have any concerns, contact us at tech@biolegend.com.
Antigen Details
- Structure
- Monomer
- Distribution
-
Mouse oligodendrocytes and neurons
- Function
- Generating amyloid beta peptide
- Interaction
- APP homologs, IL1-R2, PSGL-1, ST6Gal-1
- Ligand/Receptor
- GGA proteins
- Bioactivity
- Generating amyloid beta peptide
- Biology Area
- Cell Biology, Neurodegeneration, Neuroscience, Protein Misfolding and Aggregation
- Molecular Family
- Enzymes and Regulators
- Antigen References
-
1. Vassar R, et al. 2014. J. Neurochem. 130(1):4
2. Kandalepas P. C. and Vassar R. 2014 Curr. Alzheimer. Res. 11:441
3. Kandalepas P. C. and Vassar R. 2012 J. Neurochem. 120(Suppl. 1):55
4. Gruninger-Leitch F, et al. 2002. J. Biol. Chem. 277:4687 - Gene ID
- 23821 View all products for this Gene ID
- UniProt
- View information about BACE1 on UniProt.org
Related Pages & Pathways
Pages
Related FAQs
- Why choose BioLegend recombinant proteins?
-
• Each lot of product is quality-tested for bioactivity as indicated on the data sheet.
• Greater than 95% Purity or higher, tested on every lot of product.
• 100% Satisfaction Guarantee for quality performance, stability, and consistency.
• Ready-to-use liquid format saves time and reduces challenges associated with reconstitution.
• Bulk and customization available. Contact us.
• Learn more about our Recombinant Proteins. - How does the activity of your recombinant proteins compare to competitors?
-
We quality control each and every lot of recombinant protein. Not only do we check its bioactivity, but we also compare it against other commercially available recombinant proteins. We make sure each recombinant protein’s activity is at least as good as or better than the competition’s. In order to provide you with the best possible product, we ensure that our testing process is rigorous and thorough. If you’re curious and eager to make the switch to BioLegend recombinants, contact your sales representative today!
- What is the specific activity or ED50 of my recombinant protein?
-
The specific activity range of the protein is indicated on the product datasheets. Because the exact activity values on a per unit basis can largely fluctuate depending on a number of factors, including the nature of the assay, cell density, age of cells/passage number, culture media used, and end user technique, the specific activity is best defined as a range and we guarantee the specific activity of all our lots will be within the range indicated on the datasheet. Please note this only applies to recombinants labeled for use in bioassays. ELISA standard recombinant proteins are not recommended for bioassay usage as they are not tested for these applications.
- Have your recombinants been tested for stability?
-
Our testing shows that the recombinant proteins are able to withstand room temperature for a week without losing activity. In addition the recombinant proteins were also found to withstand four cycles of freeze and thaw without losing activity.
- Does specific activity of a recombinant protein vary between lots?
-
Specific activity will vary for each lot and for the type of experiment that is done to validate it, but all passed lots will have activity within the established ED50 range for the product and we guarantee that our products will have lot-to-lot consistency. Please conduct an experiment-specific validation to find the optimal ED50 for your system.
- How do you convert activity as an ED50 in ng/ml to a specific activity in Units/mg?
-
Use formula Specific activity (Units/mg) = 10^6/ ED50 (ng/mL)
 Login/Register
Login/Register 








Follow Us