- Clone
- D2-40 (See other available formats)
- Regulatory Status
- IVD
- Isotype
- Mouse IgG1, κ
-
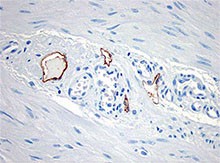
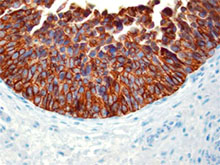
Immunohistochemistry showing lymphatic channels with staining of the endothelium. The adjacent small blood vessels show no staining of their endothelium. The section is from muscularis propria of bowel (40X). -

-
This antibody is effective in immunohistochemistry (IHC).
Clone D2-40 reacts with a O-linked sialoglycoprotein (MW 40 kD) found on lymphatic endothelium, fetal testis and on the surface of testicular germ cell tumors. Clone D2-40 (D240) has shown staining in lymphatic channel endothelium but not in the adjacent capillary.
Concentrated Format:
Cat. No. 916601 (Formerly: SIG-3730-100) [0.1 ml]
Cat. No. 916602 (Formerly: SIG-3730-1000) [1 ml]
Prediluted Formats:
Cat. No. 916603 (Formerly: SIG-3730-16) [6 ml Level 1]-ready to use for automated stainers
Cat. No. 916604 (Formerly: SIG-3730-26) [6 ml Level 2]-ready to use for manual staining
Both are ready-to-use with a Biotin-based (USA Ultra Streptavidin Detection, Cat. No. 929501) detection system.
Product Details
- Product Information
-
Intended Use:
In Vitro Diagnostic (IVD). Use in immunohistochemistry (IHC) test methods only.
The monoclonal antibody D2-40 is used for the in vitro examination of frozen or paraffin-embedded tissue sections of human bowel or esophagus samples using immunohistochemistry (IHC) methods for the qualitative identification of D2-40 Lymphatic Endothelial Marker. The clinical interpretation of any staining or its absence should be complemented by morphological studies and proper controls and should be evaluated within the context of the patient's clinical history and other diagnostic tests by a qualified pathologist.
- Reactivity
- Human
- Immunogen
- This antibody was developed using M2A.
- Formulation
- Phosphate-buffered solution with BSA + 0.1% NaN3.
- Preparation
- Ammonium sulfate precipitated TCS.
- Storage & Handling
- Store between 2°C and 8°C.
- Recommended Usage
-
Each lot of this antibody is quality control tested by immunohistochemical staining of formalin-fixed paraffin-embedded sections of human bowel and esophagus. Frozen human sections of bowel and esophagus have been verified during product development.
The optimal working dilution should be determined for each specific assay condition.
• IHC: ≥1:40 (concentrated format) for Biotin based detection systems such as USA Ultra Streptavidin Detection (Cat. No. 929501).
Tissue Sections: Formalin-fixed, paraffin-embedded tissues, frozen sections
Pretreatment: Not required
Incubation: 60 minutes at room temperature -
Application References
(PubMed link indicates BioLegend citation) -
- Hamanaka T, et al. 2011. Invest. Ophthalmol. Vis. Sci. 52:8849. (IHC) PubMed
- Choi WW, et al. 2005. Mod Pathol. 18:143. PubMed
- Chu AY, et al. 2005. Mod Pathol. 18:105.
- Dumoff KL, et al. 2005. Mod Pathol. 18:97.
- Galambos C, Nodit L. 2005. Ped Dev Pathol. 8:181.
- Fogt F, et al. 2004. Oncol Rep. 11:47.
- Fogt F, et al. 2004. Int J Molec Med. 13:681.
- RRID
-
AB_2565185 (BioLegend Cat. No. 916604)
AB_2565182 (BioLegend Cat. No. 916601)
AB_2565184 (BioLegend Cat. No. 916603)
AB_2565183 (BioLegend Cat. No. 916602)
- Disclaimer
-
WARNINGS AND PRECAUTIONS
- Use appropriate personal protective equipment and safety practices per universal precautions when working with this reagent. Refer to the reagent safety data sheet.
- This antibody contains sodium azide. Follow federal, state and local regulations to dispose of this reagent. Sodium azide build-up in metal wastepipes may lead to explosive conditions; if disposing of reagent down wastepipes, flush with water after disposal.
- All specimens, samples and any material coming in contact with them should be considered potentially infectious and should be disposed of with proper precautions and in accordance with federal, state and local regulations.
- Do not use this reagent beyond the expiration date stated on the label.
- Do not use this reagent if it appears cloudy or if there is any change in the appearance of the reagent as these may be indication of possible deterioration.
Related FAQs
Customers Also Purchased
Compare Data Across All Formats
This data display is provided for general comparisons between formats.
Your actual data may vary due to variations in samples, target cells, instruments and their settings, staining conditions, and other factors.
If you need assistance with selecting the best format contact our expert technical support team.
-
D2-40 Lymphatic Endothelial Marker Monoclonal Antibody
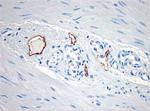
Immunohistochemistry showing lymphatic channels with stainin... 

-
Purified anti-Podoplanin (Lymphatic Endothelial Marker)
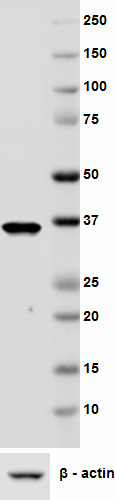
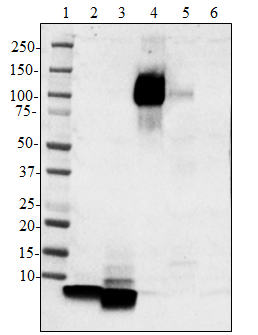
Western blot analysis of cell lysates from HeLa using Podopl... 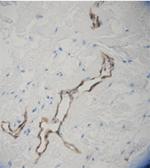
IHC staining of purified anti-Podoplanin (Lymphatic Endothel... 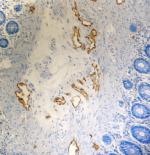
IHC staining of purified anti-Podoplanin (Lymphatic Endothel... 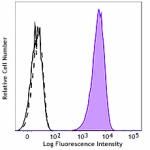
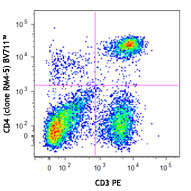
LN319 cells (positive control, filled histogram), or HeLa ce... 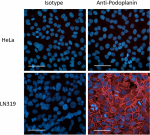
HeLa cells (negative control, top panels), or LN319 cells (p... -
Alexa Fluor® 594 anti-Podoplanin/Lymphatic Endothelial Marker
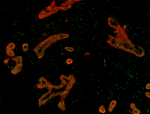
Human paraffin-embedded breast tissue slices were prepared ... -
Alexa Fluor® 647 anti-Podoplanin (Lymphatic Endothelial Marker)
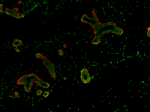
Human paraffin-embedded breast tissue slices were prepared w...
 Login / Register
Login / Register 






Follow Us