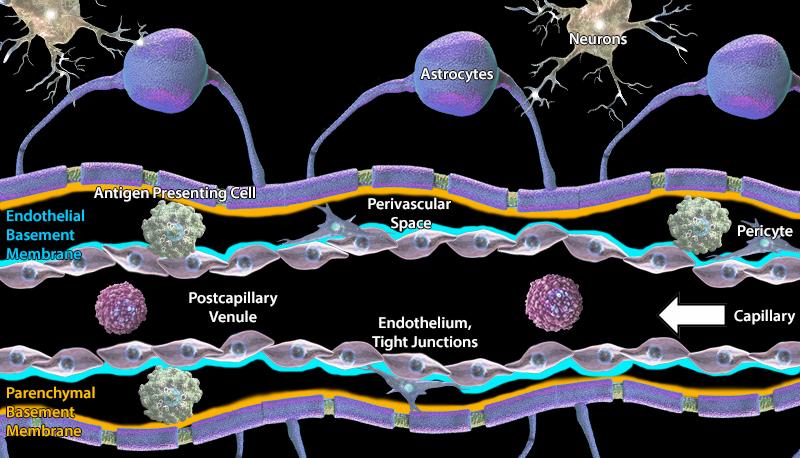
The blood-brain barrier (BBB) is a unique vasculature structure at the interface between the blood circulation and the central nervous system (CNS). This barrier is formed at the level of CNS microvessels, capillaries and post-capillary venules, and tightly regulates the passage of molecules and peripheral immune cells into the CNS. This selective BBB permeability renders the CNS parenchymal tissue an immunologically privileged site.
The BBB is composed of highly specialized endothelial cells (ECs) that establish continuous intercellular tight junctions (TJs) sealing the paracellular space. ECs demonstrate low pinocytotic activity, which coupled with TJs, limits the diffusion of molecules between and across the EC layer. In addition, ECs express low levels of leukocyte adhesion molecules preventing peripheral immune cell infiltration into the healthy CNS. ECs also create an endothelial basement membrane where pericytes can be embedded. Pericytes ensheath the outer surface of the microvessels and contribute to BBB integrity and function by regulating vessel stability, diameter and blood flow. This barrier is further reinforced by the glia limitans, which is a thin barrier composed of astrocytes and astrocytic endfeet associated with the parenchymal basement membrane. At the CNS capillaries, the endothelial and parenchymal basement membranes fuse to create one basement membrane. At the post-capillary venules, these membranes remain separate and create the inner and outer limits of cerebrospinal fluid (CSF)-filled perivascular space where antigen presenting cells can be found.
 Login / Register
Login / Register 






Follow Us