Precision Count Beads™ Protocol and Applications
Introduction
Precision Count Beads™ are designed for counting the absolute number of cells in a complex mix population and other particles by flow cytometry. Precision Count Beads™ are excited by a variety of lasers including violet (405nm), blue (488nm), yellow/green (562nm), and red (633nm).
Protocol Steps
Sample Preparation
- Perform sample preparation and staining as usual. Some examples of sample that may be used are:
- Cell lines
- Whole blood
- PBMC
- Cells from migration assays
Tip: Sample handling such as centrifugation, decanting, or transferring between different tubes can significantly affect cell counts and should be kept to a minimum - Vigorously vortex the Precision Count Beads™ bottle for 30-40 seconds to ensure complete mixing and break up of aggregates that may occur during storage.
- Add 100µl of Precision Count Beads™ per sample. Accurate pipetting is crucial at this stage. We recommend using reverse pipetting to ensure pipetting of an accurate amount of beads.
Cytometer Set Up
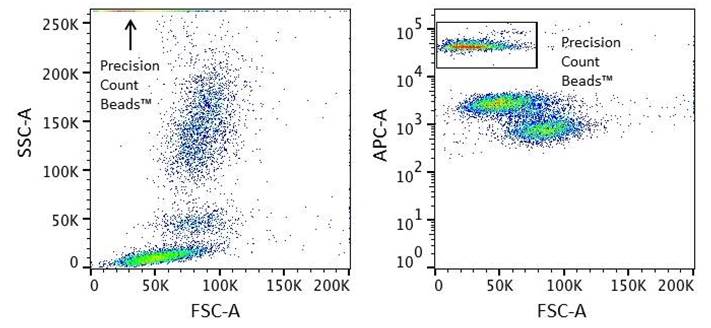
- Set up the FSC and SSC channels for the cells of interest as recommended or previously defined, ensuring that the FSC threshold is not too high. Precision Count Beads™ have a high SSC and low FSC profile. Thus, SSC should be adjusted in such a way that the cells are optimally visible. This can be done by acquiring a sample of Precision Count Beads™ alone and compare to the cells profile. Adjust compensation for the samples if needed.
- Precision Count Beads™ are highly fluorescent in all channels. Make sure to adjust the voltage to optimally detect the Precision Count Beads™ in at least one fluorescent channel.
- Acquire samples on a flow cytometer, gently vortexing every sample prior to acquisition to ensure adequate suspension of the cell and bead populations.
Data Analysis
- Data analysis should be performed as usual to identify the cell population(s) of interest.
- In an ungated plot of the same sample, Precision Count Beads™ can be visualized using one or two fluorescent parameters and a gate set around the bead population.
- The bead count and cell count can be obtained using the statistics function from the software. Absolute counts can then be calculated using formulas provided in the example below.
Example for using Precision Count Beads™ in a lyse no wash whole blood assay
1. Dilute antibody or antibodies at recommended concentration(s).
2. Add 100µL of fresh EDTA blood and incubate for 15 minutes in the dark at room temperature.
3. Slowly vortex while adding 900µL of BioLegend’s RBC lysis/fixation (1X) solution to each tube.
4. Incubate for 15 minutes in the dark.
5. Vortex Precision Count Beads™ to ensure the beads are in a homogeneous suspension.
6. Add 100µL of Precision Count Beads™ using a reverse pipetting method for improved accuracy.
7. Acquire samples on a flow cytometer.
Figure 1. It is easier to visualize Precision Count Beads™ using at least one fluorescent channel as a parameter.
8. If the volumes of the sample and the Precision Count Beads™ are the same (1:1), then the absolute cell count can be determined using the following formula: 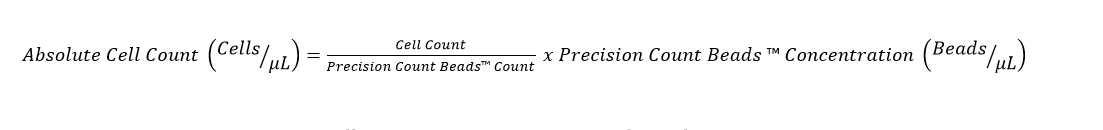
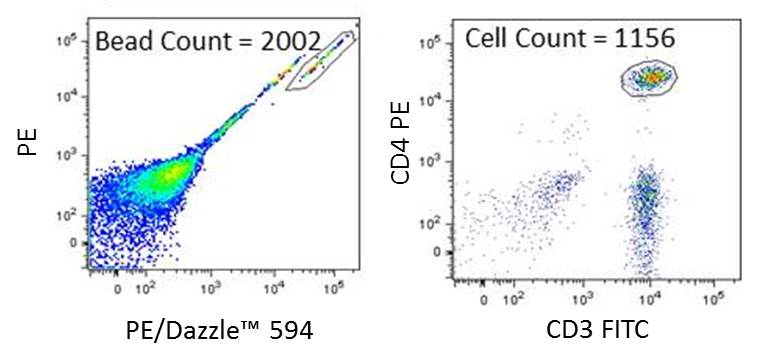
Figure 2. Human peripheral blood lymphocytes were stained with CD3 FITC/CD4 PE/CD45 PerCP cocktail (BioLegend Cat #368302) using a lyse/no wash method. Precision Count Beads™ were used as recommended. Bead count and CD3+CD4+ cell count was determined by gating on beads and cells as depicted in Figure 2. Cells were gated on lymphocytes, based on their FSC vs SSC profile, and CD45+.
In this example, the absolute number of cells can be calculated as follows:
Precision Count Beads™ number = 2002 Cell number = 1156 Precision Count Beads per µl = 1000
Absolute Cell Count = (1156/2002) X 1000 = 577 CD3+CD4+ cells/µL
9. If the relative proportion of the sample and the Precision Count Beads™ is different (not 1:1), then the absolute count can be determined as follows:
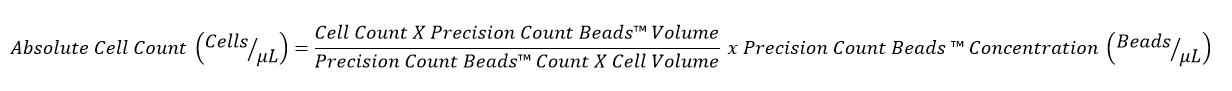
 Login/Register
Login/Register 







Follow Us