MojoSort™ Mouse CD11c Nanobeads Column Protocol
Introduction
BioLegend MojoSort™ nanobeads work in commonly used separation columns, based on our internal research as well as validation by external testing by academic labs. This simple protocol consists of following the MojoSort™ protocol to label the cells with pre-diluted MojoSort™ reagents and using the columns as indicated by the manufacturer.
Note: Due to the properties of our beads, it may be possible to use far fewer beads than with other commercial suppliers. We recommend a titration to find the best dilution factor. However, as a general rule, dilutions ranging from 1:3 to 1:20 for the Nanobeads can be used. Please contact BioLegend Technical Service (tech@biolegend.com) if further assistance is needed.
Important Note
MojoSort™ magnetic particles can be used with other commercially available magnetic separators, both free standing magnets and column-based systems. Because MojoSort™ protocols are optimized for the MojoSort™ separator, the protocols may need to be adjusted for other systems. Please contact BioLegend Technical Service (tech@biolegend.com) for more information and guidance. We do not recommend using MojoSort™ particles for BD’s IMag™ or Life Technologies’ DynaMag™.
Sample Preparation: Enzymatic digestion of mouse spleen is recommended to achieve the highest purity and yield of CD11c+ cells. There are several protocols published that can be applied. As a general guideline, cut mouse spleen into pieces and incubate in 0.5 mg/ml Collagenase for 30 to 60 minutes at room temperature or 37°C. Place the tube in a rocking platform with continuous agitation or gently pipette every 10 minutes. Alternatively, inject 1 ml of enzymes solution in the uncut organ. Force the tissue through a 70 µm filter to prepare a single cell suspension, and wash with complete media. Resuspend cells in 0.1 mg/ml DNase 1 solution and incubate at room temperature for 10 minutes. Again, filter cells through a 70 µm filter and wash with complete media. Resuspend in complete media or MojoSort™ Buffer and keep on ice until ready to use.
Protocol Steps
- Prepare cells from your tissue of interest or blood without lysing erythrocytes.
- In the final wash of your sample preparation, resuspend the cells in MojoSort™ Buffer by adding up to 4 mL in a 5 mL (12 x 75 mm) polypropylene tube.
Note: Keep MojoSort™ Buffer on ice throughout the procedure. - Filter the cells with a 70 µm cell strainer, centrifuge at 300xg for 5 minutes, and resuspend in a small volume of MojoSort™ Buffer. Count and adjust the cell concentration to 1 x 108 cells/mL by adding MojoSort™ Buffer.
- Aliquot 100 µL (107 cells) into a new tube. Add 10 µL of TruStain FcX (anti-mouse CD16/32 antibody), mix well and incubate at room temperature for 10 minutes. Scale up the volume accordingly if separating more cells. For example, if the volume of Human TruStain FcX™ for 1x107cells is 10 µL, add 100 µL for 1 x 108 cells. When working with less than 107 cells, use indicated volumes for 107cells.
- Resuspend the beads by vortexing, maximum speed, 5 touches. Add 10 µL of Antibody Nanobeads. Mix well and incubate on ice for 15 minutes. Scale up the volume accordingly if separating more cells.
- Wash the cells by adding MojoSort™ Buffer up to 4 mL. Centrifuge the cells at 300xg for 5 minutes.
- Discard the supernatant.
- Resuspend cells in the appropriate amount of MojoSort™ Buffer and proceed to separation. At least 500 µL is needed for column separation.
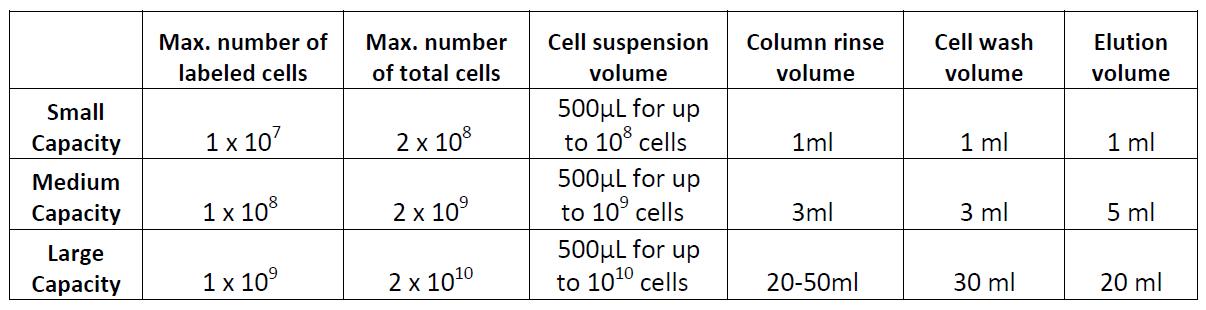
Note: There are several types of commercially available columns, depending on your application. Choose the one that fits best your experiment:
Columns:
Example of magnetic separation with medium capacity columns:
- Place the column in a magnetic separator that fits the column.
- Rinse the column with 3 mL of cell separation buffer.
- Add the labeled cell suspension to the column through a 30 µm filter and collect the fraction containing the unlabeled cells.
- Wash the cells in the column 3 times with 3 mL of buffer and collect the fraction containing the unlabeled cells. Combine with the collected fraction from step 3. These cells may be useful as controls, to monitor purity/yield, or other purposes.
- Take away the column from the magnet and place it on a tube. Then add 5 mL of buffer and flush out the magnetically labeled fraction with a plunger or supplied device. These are the positively isolated cells of interest; do not discard. To increase the purity of the magnetically labeled fraction repeat the isolation process with a new, freshly prepared column.
Data
Flow cytometry. High purity and yield. “After Isolation” plot shows purified population of interest using pre-diluted MojoSort™ reagents in separation columns.
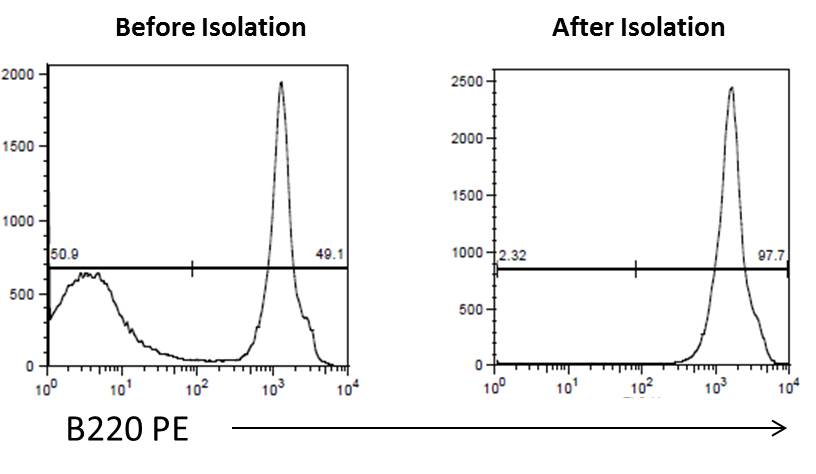
| Kit | Purity | Yield |
|---|---|---|
| Mouse CD19 Nanobeads | 97.7% | 94% |
Electron Microscopy. MojoSort™ Nanobead-isolated CD19+ cells using columns do not display more bound beads on the cell surface (A) as compared to cells isolated with a compatible commercial product using the same columns (B). Red arrows indicate where the particles are located. Numbers indicate either 2 or 3 magnetic particles adjacent to each other. Pictures were taken at the same magnification, scale shown in B. Images are representative of 41 different cells each.
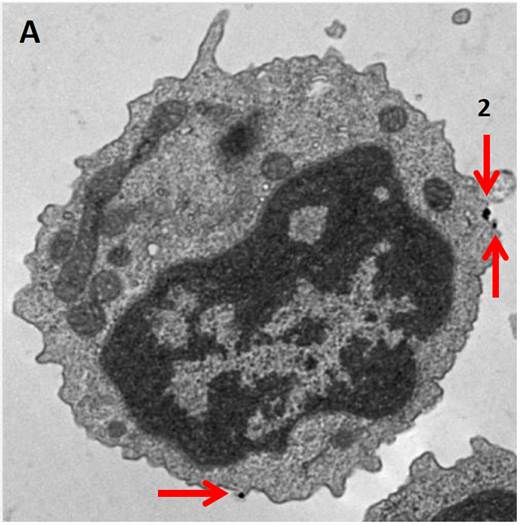 B cells isolated with MojoSort™ CD19 nanobeads using separation columns. |
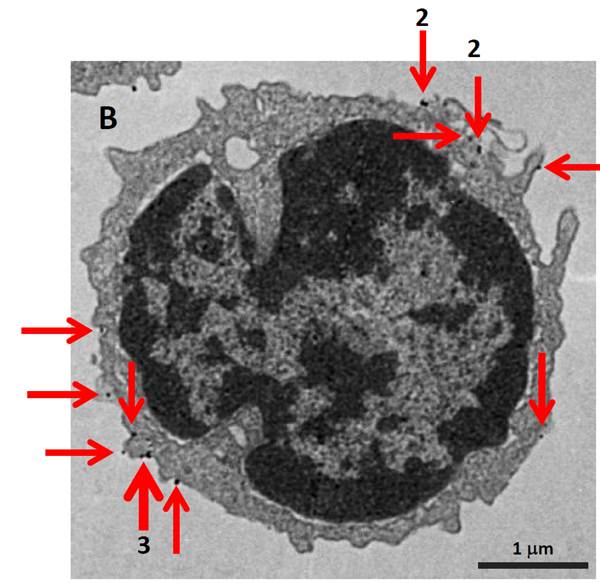 B cells isolated with competitor's CD19 magnetic beads using separation columns. |
 Login/Register
Login/Register 







Follow Us