MojoSort™ Human CD45 Nanobeads Protocol 2 - CD45 greater than 50%
Reagent List
|
Important Note
MojoSort™ magnetic particles can be used with other commercially available magnetic separators, both free standing magnets and column-based systems. Because MojoSort™ protocols are optimized for the MojoSort™ separator, the protocols may need to be adjusted for other systems. Please contact BioLegend Technical Service (tech@biolegend.com) for more information and guidance. We do not recommend using MojoSort™ particles for BD’s IMag™ or Life Technologies’ DynaMag™.
Protocol Steps
Product description and procedure summary:
If the percentage of CD45+ cells in your sample is less than 50%, please follow Human CD45 Nanobeads Protocol 1. If it is higher than 50% then please follow Human CD45 Nanobeads Protocol 2.
The cells targeted by the Nanobeads are either selected or depleted by incubating your sample with the directly conjugated magnetic particles. The magnetically labeled fraction is retained by the use of a magnetic separator. After collection of the targeted cells, downstream applications include functional assays, gene expression, phenotypic characterization, etc.
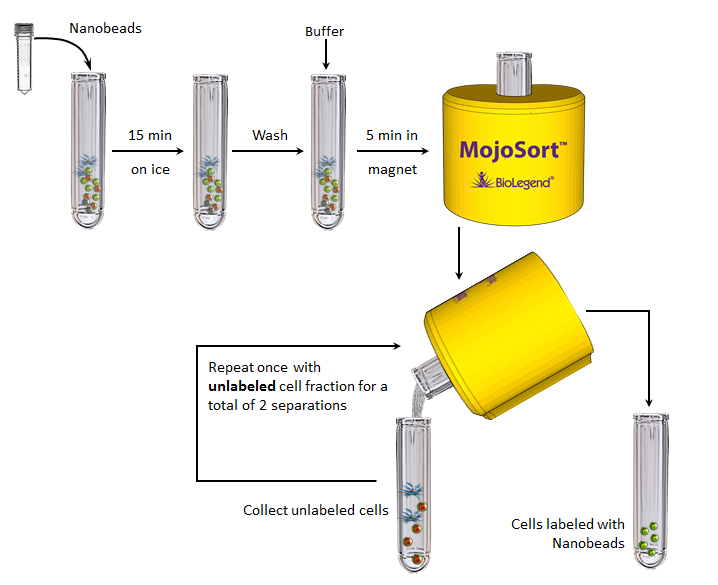
Note: This procedure is optimized for the isolation of 107 to 2 x 108cells per tube. If working with fewer than 107 cells, keep volumes as indicated for 107 cells. For best results, optimize the conditions to your specific cell number and tissue. Prepare fresh MojoSort™ Buffer solution by diluting the 5X concentrate with sterile distilled water. Scale up volumes if using 14mL tubes and Magnet, and place the tube in the magnet for 10 minutes.
- Prepare cells from your tissue of interest or blood without lysing erythrocytes. Kits for human samples have been optimized for PBMCs, please prepare the cells using a suitable method.
- In the final wash of your sample preparation, resuspend the cells in MojoSort™ Buffer by adding up to 4 mL in a 5 mL (12 x 75 mm) polypropylene tube.
Note: Keep MojoSort™ Buffer on ice throughout the procedure. - Filter the cells with a 70µm cell strainer, centrifuge at 300xg for 5 minutes, and resuspend in an appropriate volume of MojoSort™ Buffer. Count and adjust the cell concentration to 1 x 108 cells/mL.
- Aliquot 100 µL of cell suspension (107 cells) into a new tube.
- Resuspend the beads by vortexing, maximum speed, 5 touches. Add 10µL of Nanobeads, mix well and incubate on ice for 15 minutes. Scale up the volume accordingly if separating more cells. For example, add 100µL for 1 x 108 cells. When working with less than 107 cells, use indicated volumes for 107 cells.
- Add MojoSort™ Buffer up to 4mL and centrifuge the cells at 300xg for 5 minutes.
- Resuspend the cells in 3mL of MojoSort™ Buffer.
Optional: Take an aliquot before placing the tube in the magnet to monitor purity and yield. - Place the tube in the magnet for 5 minutes.
- Pour out the liquid containing the unlabeled fraction.
- Remove the tube from the magnet and resuspend the first labeled fraction in appropriate amount of buffer.
- Place the tube containing the unlabeled fraction back in the magnet for 5 minutes.
- Pour out the liquid containing the unlabeled fraction from the second magnetic incubation. These are the CD45- cells, ready to use as needed.
- Remove the tube from the magnet and use the fraction obtained in step 10 to resuspend this second labeled fraction and pool them together. These are the CD45+ cells, ready to use as needed.
Optional: Take a small aliquot to monitor purity and yield. If desired, pool the unlabeled fractions and process simultaneously with the positive labeled cells when assessing purity and yield.
Chart Protocol:
Application notes: To use this product in magnetic separation columns, a titration of the Nanobeads should be performed. Optimal concentration for magnetic separation columns is lot-specific. Please contact BioLegend Technical Service (tech@biolegend.com) for further assistance on how to use MojoSort™ Nanobeads in magnetic separation columns.
 Login/Register
Login/Register 







Follow Us