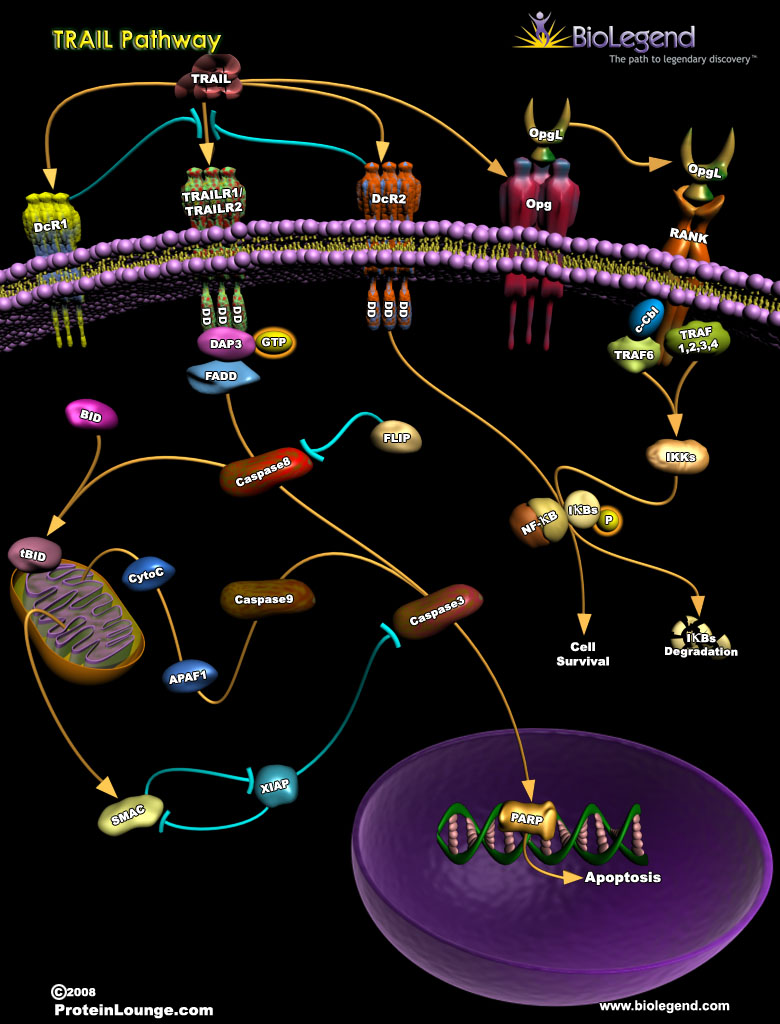
TRAIL Pathway
Tumor necrosis factor (TNF)-related apoptosis-inducing ligand (TRAIL) binds to TRAIL receptor 1 and 2 (TRAILR1/TRAILR2) on the cell surface to stimulate apoptosis, making it an important anti-tumor effector molecule. TRAILR1/2 assemble as trimers, and upon stimulation, they recruit the adaptor protein FADD to their intracellular death domains. Next, caspase 8 is activated, which can then either cleave the protein BID to initiate the mitochondrial apoptosis pathway, or directly cleave caspase 3. The TRAIL pathway can be inhibited by several mechanisms, which are used by tumor cells to escape TRAIL-mediated cell death. DcR1 lacks death domains and can compete with TRAILR1/2 for TRAIL binding, resulting in a dampened apoptotic signal. Binding of TRAIL to DcR2 and osteoprotegerin (OPG) results in stimulation of NF-κB activity, which promotes cell survival instead of death. Finally, FLIP is a homolog of caspase 8 that has no catalytic activity and can inhibit activation of the caspase pathway.
Click on the poster below to view the interactive version.
 Login/Register
Login/Register 







Follow Us