Sumo Pathway
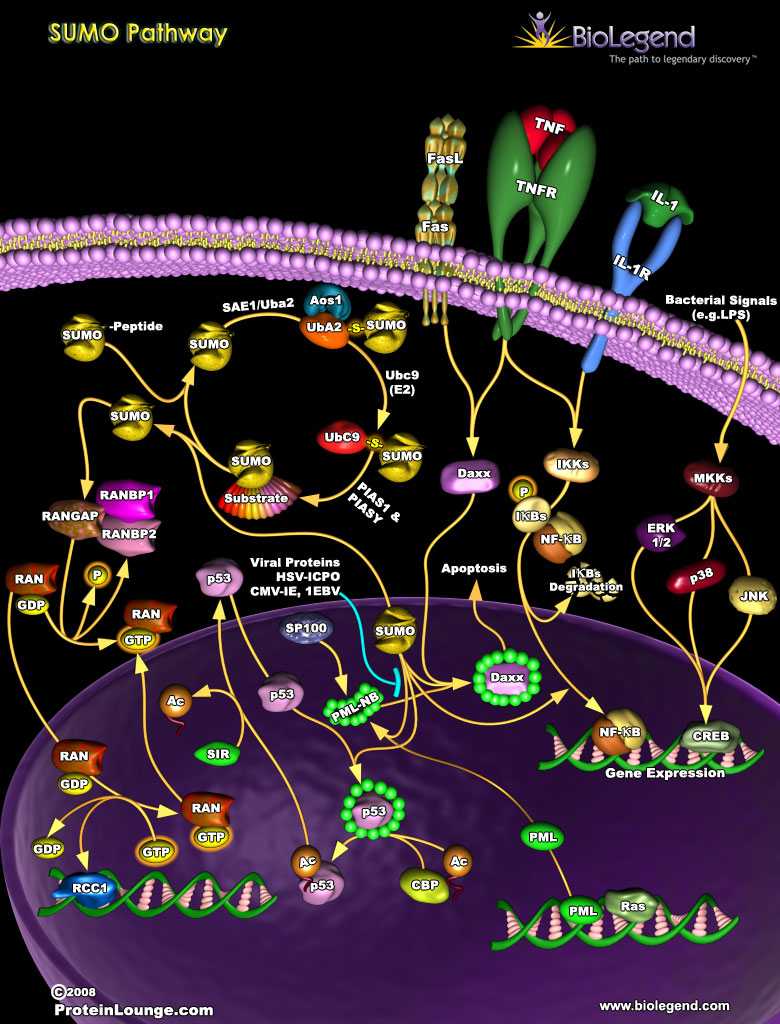
Small ubiquitin-related modifier (SUMO) proteins are attached to cellular proteins in a process called sumoylation, which is used to control functions like transcription, cell cycle progression, and DNA repair. Processing of SUMO into its mature form allows for its adenylation and activation by UBA2 (SUMO-activating enzyme subunit 2), and then its conjugation to a substrate by UbC9 (ubiquitin-like conjugating enzyme 9). One of the first known substrates of SUMO was RanGAP, which activates Ran GTPase upon sumoylation. Activation of Ran is critical for transport of proteins across the nuclear pore complex (NPC). Other pathways regulated by SUMO include p53-induced apoptosis and transcription of pro-inflammatory genes by NF-κB. SUMO activates p53 transcriptional activity by direct modification, while it negatively regulates NF-κB activity by blocking inhibitor of κB (IκB) from ubiquitination and subsequent degradation.
Click on the poster below to view the interactive version.
 Login/Register
Login/Register 







Follow Us