- Clone
- 3H6.H7 (See other available formats)
- Regulatory Status
- RUO
- Other Names
- Microtubule-associated protein tau, paired helical filament tau, PHF tau, neurofibrillary tangle protein
- Previously
-
Covance Catalog# MMS-5065
- Isotype
- Mouse IgG1, κ
- Ave. Rating
- Submit a Review
- Product Citations
- publications
-
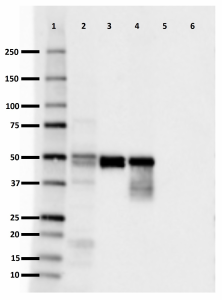
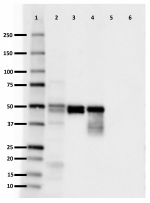
Western blot of purified anti-0N Tau antibody (clone 3H6.H7). Lane 1: Molecular weight marker; Lane 2: 20 µg of human brain lysate; Lane 3: 0.1 µg of recombinant human Tau ladder protein; Lane 4: 0.5 µg of recombinant human 0N3R Tau protein; Lane 5: 0.5 µg of recombinant human 1N3R Tau protein; Lane 6: 0.5 µg of recombinant human 2N3R Tau protein. The blot was incubated with 10 µg/mL of the primary antibody overnight at 4°C, followed by incubation with HRP labeled goat anti-mouse IgG (Cat. No. 405306). Enhanced chemiluminescence was used as the detection system. -
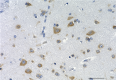
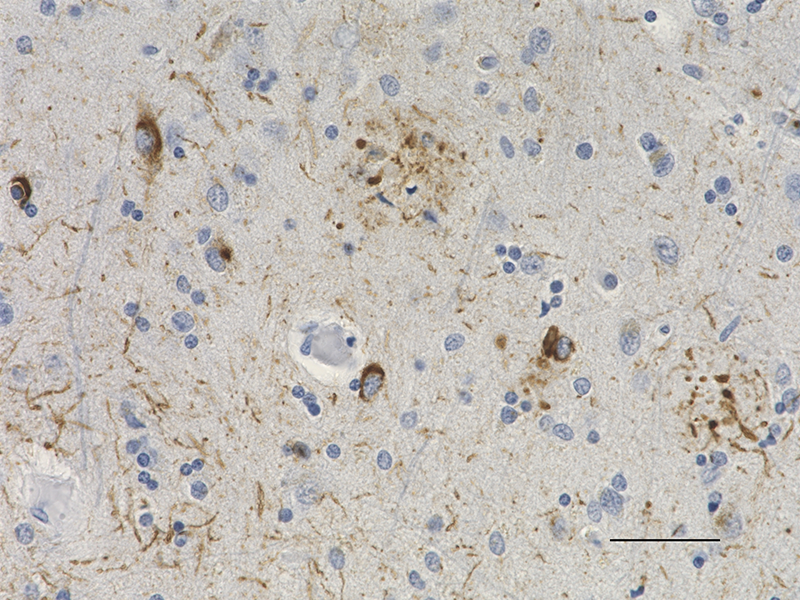
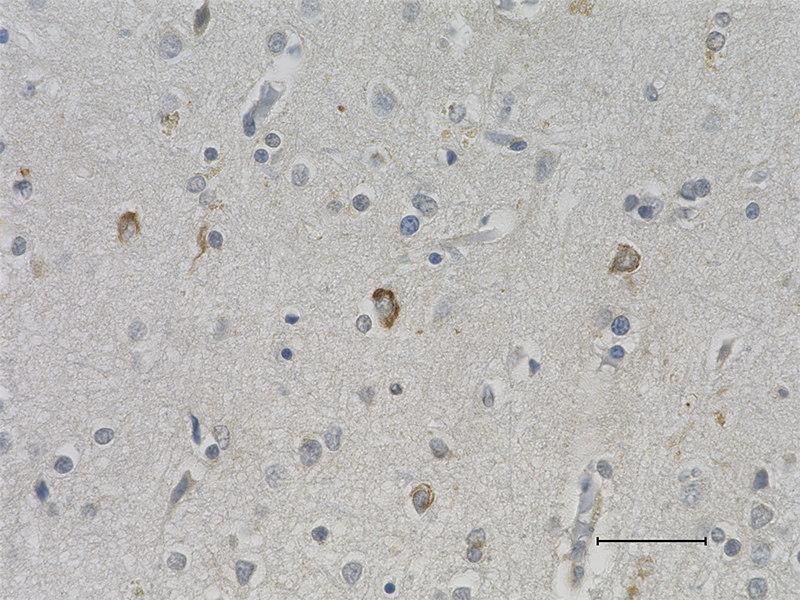
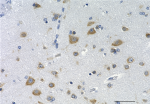
IHC staining of purified anti-0N Tau antibody (clone 3H6.H7) on formalin-fixed paraffin-embedded Alzheimer's disease human brain tissue. Following antigen retrieval using Sodium Citrate H.I.E.R., the tissue was incubated with 10 µg/ml of the primary antibody overnight at 4°C. BioLegend's Ultra-Streptavidin (USA) HRP kit (Multi-Species, DAB, Cat. No. 929901) was used for detection followed by hematoxylin counterstaining, according to the protocol provided. The image was captured with a 40X objective. Scale bar: 50 µm
| Cat # | Size | Price | Quantity Check Availability | Save | ||
|---|---|---|---|---|---|---|
| 823802 | 25 µL | 90€ | ||||
| 823801 | 100 µL | 220€ | ||||
Tau proteins are microtubule-associated protein (MAPs) which are abundant in neurons of the central nervous system, but are also expressed at very low levels in CNS astrocytes and oligodendrocytes and elsewhere. One of tau's main functions is to modulate the stability of axonal microtubules. Tau is active primarily in the distal portions of axons providing microtubule stabilization as well as flexibility. Pathologies and dementias of the nervous system such as Alzheimer's disease feature tau proteins that have become defective and no longer stabilize microtubules properly. As a result, tau forms aggregates with specific structural properties referred to as Paired Helical Filaments (PHFs) that are a characteristic of many different types of dementias, known as tauopathies.
Tau has two primary ways of controlling microtubule stability: isoforms and phosphorylation. Six tau isoforms exist in human brain tissue, and they are distinguished by the number of binding domains. Three isoforms have three binding domains and the remaining three have four binding domains. The binding domains are located in the carboxy-terminus of the protein and are positively-charged (for binding to the negatively-charged microtubule). Tau isoforms with four binding domains are better at stabilizing microtubules than those with three binding domains.
Thus, in the human brain, the tau proteins constitute a family of six isoforms with the range from 352-441 amino acids. They also differ in either zero, one or two inserts of 29 amino acids at the N-terminal part (exon 2 and 3), and three or four repeat-binding regions at the C-terminus. So, the longest isoform in the CNS has four repeats (R1, R2, R3 and R4) and two inserts (441 amino acids total), while the shortest isoform has three repeats (R1, R3 and R4) and no insert (352 amino acids total). Tau is also a phosphoprotein with 79 potential Serine (Ser) and Threonine (Thr) phosphorylation sites on the longest tau isoform. Phosphorylation has been reported on approximately 30 of these sites in normal tau proteins. Mechanisms that drive tau lesion formation in the highly prevalent sporadic form of AD are not fully understood, but appear to involve abnormal post-translational modifications (PTMs) that influence tau function, stability, and aggregation propensity.
Product Details
- Verified Reactivity
- Human
- Antibody Type
- Monoclonal
- Host Species
- Mouse
- Immunogen
- This monoclonal antibody was developed against a peptide sequence corresponding to amino acids 39-50 of the human 0N3R and 0N4R tau proteins.
- Formulation
- Phosphate-buffered solution.
- Preparation
- The antibody was purified by affinity chromatography.
- Concentration
- 1 mg/ml
- Storage & Handling
- This antibody should be handled aseptically as it is free of preservatives such as Sodium Azide. Store this antibody undiluted between 2°C and 8°C. Please note the storage condition for this antibody has been changed from -20°C to between 2°C and 8°C. You can also check the vial label or CoA to find the proper storage conditions.
- Application
-
WB - Quality tested
IHC-P - Verified - Recommended Usage
-
Each lot of this antibody is quality control tested by Western blotting. For Western blotting, the suggested use of this reagent is 2 - 10 µg per ml. For immunohistochemistry, a concentration of 10 µg/ml is suggested. It is recommended that the reagent be titrated for optimal performance for each application.
- Product Citations
-
- RRID
-
AB_2783399 (BioLegend Cat. No. 823802)
AB_2564874 (BioLegend Cat. No. 823801)
Antigen Details
- Structure
- Expected MW: 51 kD, 53 kD
- Biology Area
- Cell Biology, Neurodegeneration, Neuroscience, Protein Misfolding and Aggregation
- Molecular Family
- Tau
- Gene ID
- 4137 View all products for this Gene ID
- UniProt
- View information about 0N Tau on UniProt.org
Related Pages & Pathways
Pages
Related FAQs
Other Formats
View All 0N Tau Reagents Request Custom Conjugation| Description | Clone | Applications |
|---|---|---|
| Purified anti-0N Tau | 3H6.H7 | WB,IHC-P |
Customers Also Purchased
Compare Data Across All Formats
This data display is provided for general comparisons between formats.
Your actual data may vary due to variations in samples, target cells, instruments and their settings, staining conditions, and other factors.
If you need assistance with selecting the best format contact our expert technical support team.
 Login / Register
Login / Register 












Follow Us