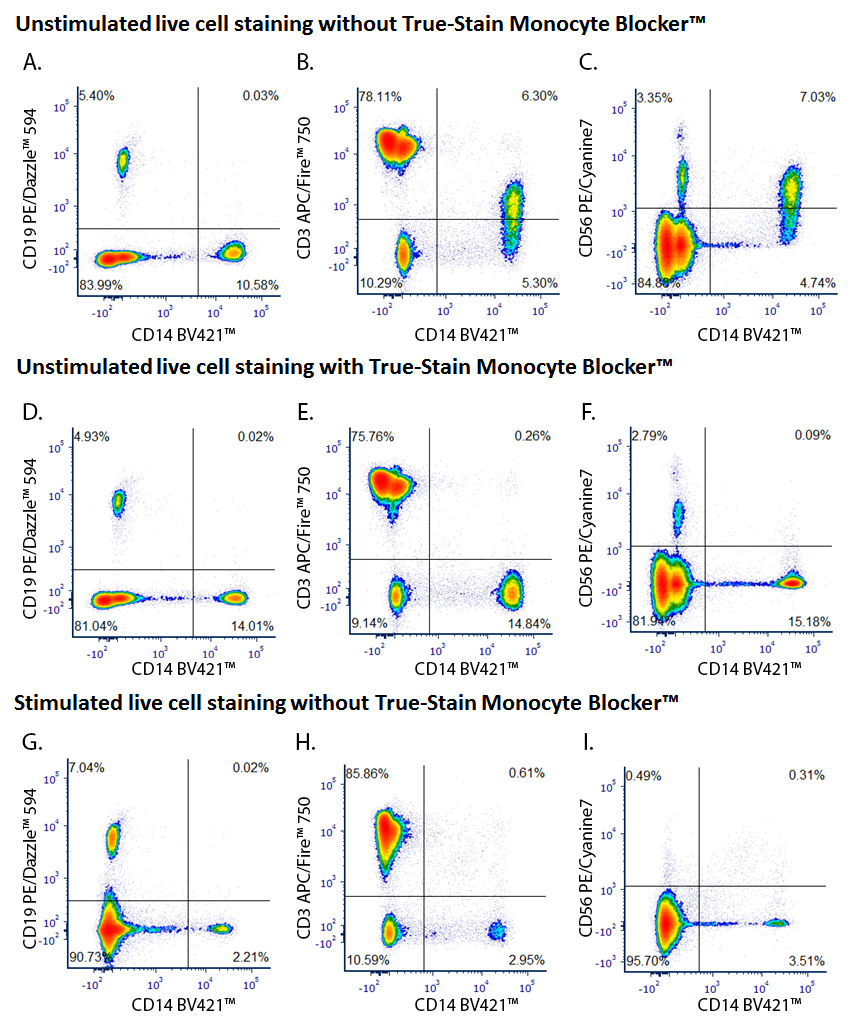
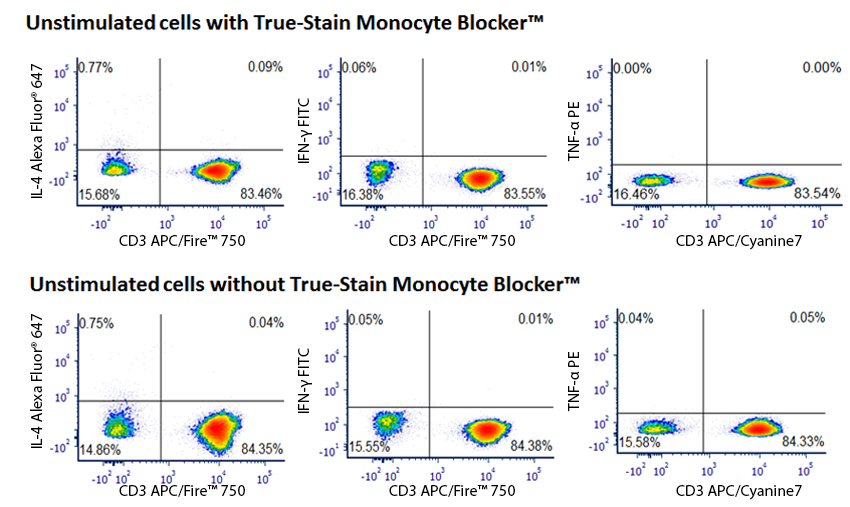
Don’t be Fooled by Monocyte Non-Specific Antibody Binding
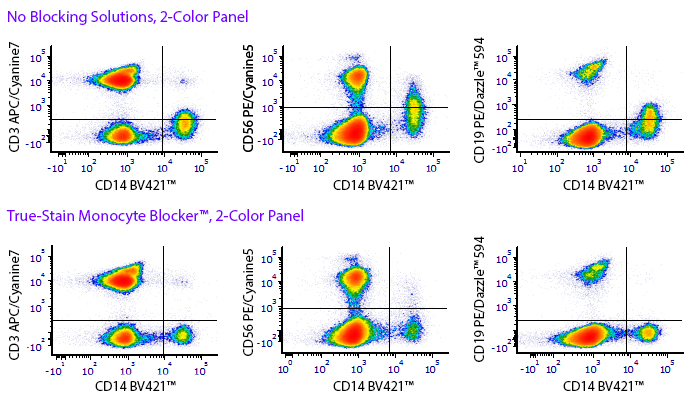
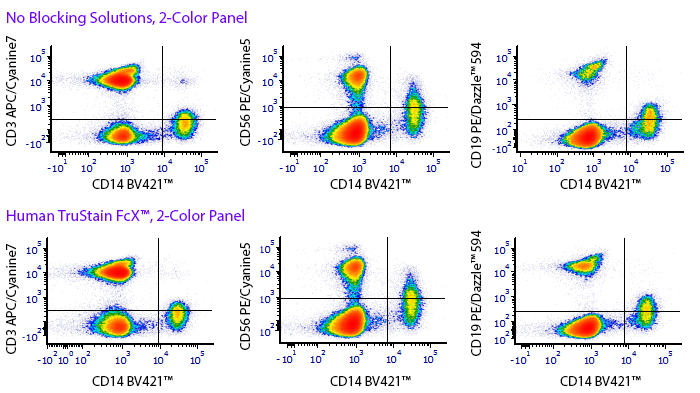
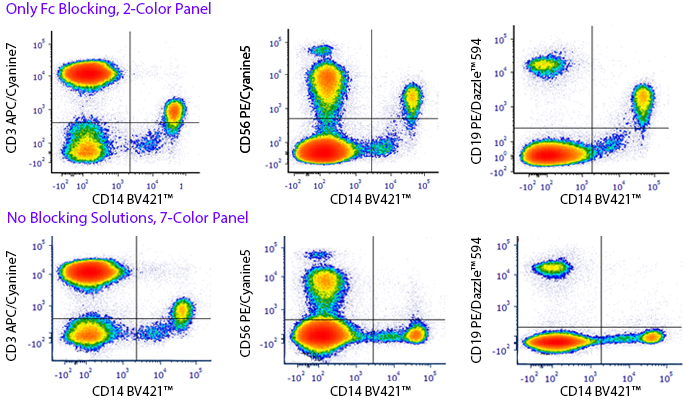
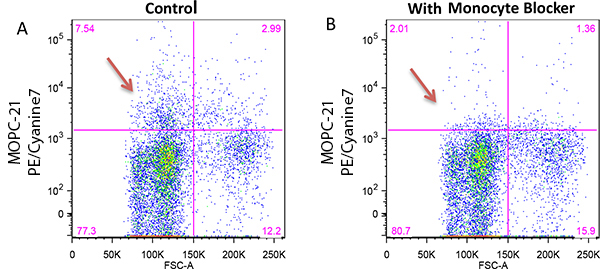
Monocytes and macrophages can exhibit non-specific binding of antibodies and fluorophores used in cell surface staining of live cells. True-Stain Monocyte Blocker™ is a non-antibody based blocking solution that has been shown to reduce non-specific fluorophore interactions without affecting the the true staining of monocytes. Stain monocytes and fully trust your results.
Download the PDF for our scientific poster on True-Stain Monocyte Blocker™.
 Login/Register
Login/Register 






Follow Us