- Other Names
- BMP2A, BMP-2A, BMP-2, Bone Morphogenetic Protein 2A
- Ave. Rating
- Submit a Review
- Product Citations
- publications
-
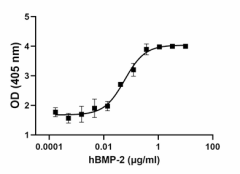
GMP recombinant human BMP-2 induces alkaline phosphatase (ALP) production in the mouse chrondrogenic cell line ATDC5 in a dose dependent manner. The ED50 for this effect is 40 – 200 ng/mL. -

Stability Testing for GMP Recombinant Human BMP-2. Human BMP-2 was aliquoted in 4mM HCl. One aliquot was frozen and thawed four times (4x Freeze/Thaw) and compared to the control that was kept at 4°C (Control). The samples were tested for their ability to induce alkaline phosphatase (ALP) production in the mouse chrondrogenic cell line ATDC5 in a dose dependent manner. The ED50 for this effect is 40 – 200 ng/mL.
| Cat # | Size | Price | Quantity Check Availability | Save | ||
|---|---|---|---|---|---|---|
| 767316 | 100 µg | 1760€ | ||||
| 767314 | 25 µg | 688€ | ||||
Bone morphogenetic protein 2 (BMP-2) belongs to the TGF-β superfamily. Like other members of BMP family, BMP-2 is synthesized as an inactive propeptide precursor which dimerizes and then, it is further processed into mature form by proprotein convertases (PCs). Some evidence indicated that PC5/6A and Factor VII-activating protease (FSAP) are involved in maturation of BMP-2. Mature BMP-2 is a 26 kD protein composed of 114 amino acids, forming three intramolecular and one intermolecular disulfide bond. BMP-2 forms homodimer or heterodimer with other BMP proteins, including BMP-4, BMP-6 and BMP-7. BMP-2 signal through heterodimeric serine/threonine kinase receptors composed of a type I (BMPR1a/ALK3, BMPR1b/ALK6, ActRIa/ALK2) and a type II (BMPR2, ACVR2a/ActRIIA, ACVR2b/ActRIIB). BMP-2 binds to the type I receptor with high affinity, in turn recruiting the type II receptor. BMP-2 stimulation initiates receptor shutdown, leading to receptor clustering and activation of the downstream signaling. BMP-2 signals via canonical Smad or other downstream kinase, such as p38 and JNK in a context-dependent manner. BMP-2 is involved in several processes, including cartilage and bone formation, differentiation, and embryogenesis. BMP-2 induces osteogenic differentiation in human mesenchymal stem cells and myogenic cells. BMP-2 induces cartilage repair and remodeling by stimulating chondrocyte proliferation and expression of matrix proteins. BMP-2/BMP-7 heterodimer is more potent in the induction of bone formation in vivo than BMP-2 homodimer. BMP-2-deficiency leads to embryonic lethality with abnormal cardiac development, malformation of the amnion/chorion, severe chondrodysplasia, and defects in myocardial patterning, suggesting that BMP-2 mediates organ morphogenesis. Noggin is an antagonist that can reverse BMP-2-mediated effect. Noggin exppression is induced by BMP-2 in osteoblasts as a negative feedback loop. In addition, BMP-2 stimulates epithelial to mesenchymal cell transformation through TGF-β type III receptor.
Product DetailsProduct Details
- Source
- Human BMP-2, amino acid (Ala284-Arg396) (Accession: # P12643) was expressed in E.coli.
- Molecular Mass
- The 113 amino acid recombinant protein has a predicted molecular mass of approximately 12.8 kD. The DTT-reduced and non-reduced protein migrates at approximately 13 and 26 kD respectively by SDS-PAGE. The predicted N-terminal amino acid is Ala.
- Purity
- > 95%, as determined by Coomassie stained SDS-PAGE.
- Formulation
- 0.1 µm filtered protein solution is in 4 mM HCl
- Endotoxin Level
- Less than 0.1 EU per µg cytokine as determined by the LAL method.
- Concentration
- 25 µg and 100 µg sizes are bottle at 0.5 mg/mL
- Storage & Handling
- Unopened vial can be stored between 2°C and 8°C for up to 2 weeks, at -20°C for up to six months, or at -70°C or colder until the expiration date. For maximum results, quick spin vial prior to opening. The protein can be aliquoted and stored at -20°C or colder. Stock solutions can also be prepared at 50 - 100 µg/mL in appropriate sterile buffer, carrier protein such as 0.2 - 1% endotoxin-free BSA or HSA can be added when preparing the stock solution. Aliquots can be stored between 2°C and 8°C for up to one week or stored at -20°C or colder for up to 3 months. Avoid repeated freeze/thaw cycles.
- Activity
- Human BMP-2 induces alkaline phosphatase (ALP) production in the mouse chrondrogenic cell line ATDC5 in a dose dependent manner. The ED50 for this effect is 40 – 200 ng/mL.
- Application
-
Bioassay
- Application Notes
-
BioLegend carrier-free recombinant proteins provided in liquid format are shipped on blue-ice. Our comparison testing data indicates that when handled and stored as recommended, the liquid format has equal or better stability and shelf-life compared to commercially available lyophilized proteins after reconstitution. Our liquid proteins are verified in-house to maintain activity after shipping on blue ice and are backed by our 100% satisfaction guarantee. If you have any concerns, contact us at tech@biolegend.com.
- Disclaimer
-
GMP Recombinant Proteins. BioLegend GMP recombinant proteins are manufactured in a dedicated GMP facility and compliant with ISO 13485:2016. For research or ex vivo cell processing use. Not for use in diagnostic or therapeutic procedures. Our processes include:
- Batch-to-batch consistency
- Material traceability
- Documented procedures
- Documented employee training
- Equipment maintenance and monitoring records
- Lot-specific certificates of analysis
- Quality audits per ISO 13485:2016
- QA review of released products
BioLegend GMP recombinant proteins are manufactured and tested in accordance with USP Chapter 1043, Ancillary Materials for Cell, Gene and Tissue-Engineered Products and Ph. Eur. Chapter 5.2.12.
Antigen Details
- Structure
- Disulfide-linked homodimer
- Distribution
-
Abundant in lung, spleen and colon in human, low expression but detectable in heart, kidney, brain, liver, skeletal muscle, pancreases, placenta, prostate, ovary, and small intestine
- Function
- BMP-2 is involved in osteogenesis, cartilage repair, and organogenesis
- Interaction
- Chondroblast, osteoblast, proprotein convertases, BMP family members
- Ligand/Receptor
- BMP receptor type IA (BMPR1a/ALK3), type IB (BMPR1b/ALK6), Activin receptor type IA (ACVR1a/ActRIa/ALK2) and BMP receptor type II (BMPR2), Activin receptors type IIA (ACVR2a/ActRIIA), type IIB (ACVR2b/ActRIIB)
- Bioactivity
- BMP-2 induces alkaline phosphatase in ATDC5 mouse chondrogenic cells.
- Cell Type
- Embryonic Stem Cells
- Biology Area
- Angiogenesis, Cell Biology, Neuroscience, Stem Cells, Synaptic Biology
- Molecular Family
- Cytokines/Chemokines, Growth Factors
- Antigen References
-
- Bragdon B, et al. 2011. Cell Signal 23: 609.
- Israel DI, et al. 1992. Growth Factor. 7: 139.
- Lee SN, et al. 2015. Am. J. Respir. Cell Mol. Biol. 52: 749.
- Roedel EK, et al. 2013. J. Biol. Chem. 288, 7193.
- Nohe A, et al. 2002. J. Biol. Chem. 277: 5330.
- Zhou AJ, et al. 2012. Growth Factors 30: 267.
- Miyazono K, et al. 2010. J. Biochem. 147: 35.
- Nohe A et al. 2004. J. Cell Sci. 118: 643.
- Hay E, et al. 2001. J. Biol. Chem. 276: 29028.
- Wozney JM, et al. 1988. Science 242: 1528.
- Ryoo HM, et al. 2006. Gene 366: 51.
- Lavery K, et al. 2008. J. Biol. Chem. 283: 20948.
- Israel DI, et al. 1996. Growth Factors 13: 291.
- Mundy G, et al. 1999. Science 286: 1946.
- De Luca F et al. 2001. Endocrinology 142: 430.
- Blaney Davidson EN, et al. 2007. Arthritis Res. Ther. 9: R102.
- Zhang H, Bradley A. 1996. Development 122: 2977.
- Pera MF, et al. 2004. J. Cell Sci. 117: 1269.
- Wang RN, et al. 2014. Genes Dis. 87-105.
- Kirkbride KC, et al. 2008. J. Biol. Chem. 283: 7628.
- Gene ID
- 650 View all products for this Gene ID
- UniProt
- View information about BMP-2 on UniProt.org
 Login / Register
Login / Register 













Follow Us