Using Beads for Sample Compensation
| Flow cytometry has continuously developed over the years. This includes an increasing amount of colors that can be detected, which expands the numbers of parameters collected simultaneously, allowing for the study of many cell types in a mixed population sample. | |
| The explosion of colors has come hand in hand with the development of new fluorophores, including tandem dyes. So, with the increased complexity in multiparameter flow cytometry, a good way to perform compensation in your instrument is by using beads. We will discuss the advantages (and some of the drawbacks) of these products. | |
| But before we discuss specific products, what are these compensation beads, and how do they work? They are microparticles that have rather homogeneous properties, with relatively constant autofluorescence (ideally low) and are designed to bind the antibodies you will use in your flow cytometry experiment. In that way, you can always have a positive control for all your conjugates. At the same time, you can have beads that do not bind any antibody; that will be your negative control. | |
| Convenience for use with tandem dyes Tandem dyes are complex molecules and their properties can vary substantially. That is why it is not advisable to use different antibodies, even if they are conjugated to the same tandem, for compensation and staining your sample. Their spectral characteristics may also vary from manufacturer to manufacturer, which makes them an important factor in your experiment (check our tandem dyes page). This is even more important when the tandem is conjugated to an antibody that binds a relatively rare molecule, which brings me to the next point. |
|
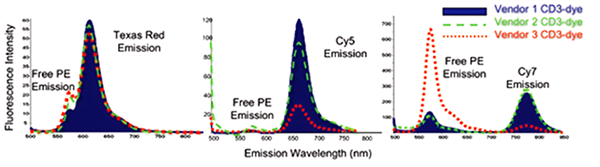 From left to right, plots show performance of PE-Texas Red®, PE-Cy5 and PE-Cy7 conjugated to CD3 from 3 different vendors. Click to read the paper. |
|
| Convenience for use with lowly expressed or rare markers Some markers are expressed by only a small population within your sample, or are dimly expressed. In both cases, you can use the beads and still generate a good positive signal. |
|
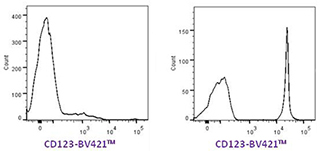 |
The left histogram shows human PBMCs stained with anti-CD123 BV421™ as a single color control. The right histogram shows the same antibody applied to OneComp eBeads from eBioscience. The positive signal is very weak in the left plot whereas the separation in the right plot is very good. |
| Easier compensation for complex multicolor experiments and convenience when working with precious samples | |
| The more colors you have, the more complex your set up will become. Beads allow you to have a homogeneous system where the fluorescence between the positive and the negative population does not depend on the abundance of the antigen, or the cell type. In addition, you don't have to use cells for compensation. This is very convenient when doing many colors, as you will save cells for each individual single color control. | |
| There are a few things to take into account when using beads though: For example, BD's beads come in three different formats, anti-mouse, anti-rat and anti-rat/hamster, so keep that in mind if you have several isotypes in your multicolor staining. In addition, some fluorophores are not quite compatible with the beads, such as BD Horizon™ V500 and AmCyan. They also only bind to antibodies with kappa light chains, so antibodies with lambda light chains are not compatible. Some other beads will come already conjugated to fluorophores, which makes them easy to use, but not very versatile, as you can't use the exact same antibodies you will have in your experiment, and are restricted to just a few colors. In addition, the products listed above are not designed for dyes that discriminate dead cells (amine reactive dyes). But there are ways to circumvent this, just write us an email to find out (mtam@biolegend.com), or wait for an upcoming post on this! Contributed by Miguel Tam, Ph.D. Note: Brilliant Violet™ is a trademark of Sirigen Group Ltd and BD Horizon™ is a trademark of Becton, Dickinson, and Company. |
|
 Login / Register
Login / Register 






Follow Us