Tau protein is expressed abundantly in neurons and has a number of functions in the central nervous system. Tau binds to microtubules and leads to their assembly, stabilization, and maintenance. Tau also regulates motor-driven axonal transport. Phosphorylation of tau is a common post-translational modification that plays an important role in the solubility, localization, and function of tau. Conformational changes in tau have been linked to excessive phosphorylation of this protein, and decreased microtubule binding and stability. Hyperphosphorylated tau has a propensity to accumulate and form protein aggregates in neurons. These aggregates eventually form intracellular filamentous inclusions, known as neurofibrillary tangles (NFTs), that are detected biochemically and immunohistologically in neurodegenerative disorders termed Tauopathies. Hyperphosphorylated and aggregated tau interferes with normal neuronal function, such as microtubule dynamics, and ultimately leads to neurodegeneration.
 Login/Register
Login/Register 



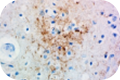 Tau exists in six isoforms that are differentially expressed during development. These isoforms are distinguished by the number of tubulin binding domains, 3 (3R) or 4 (4R), in the C-terminus and by one (1N), two (2N), or no (0N) inserts in the N-terminus. Clone A16103A recognizes human 3R and 4R isoforms and show minimal to no reactivity with murine tau protein.
Tau exists in six isoforms that are differentially expressed during development. These isoforms are distinguished by the number of tubulin binding domains, 3 (3R) or 4 (4R), in the C-terminus and by one (1N), two (2N), or no (0N) inserts in the N-terminus. Clone A16103A recognizes human 3R and 4R isoforms and show minimal to no reactivity with murine tau protein.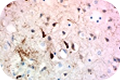 Clone A16097F also detects human 3R and 4R isoforms with minimal murine cross-reactivity, and can be used for both IHC and western blotting.
Clone A16097F also detects human 3R and 4R isoforms with minimal murine cross-reactivity, and can be used for both IHC and western blotting. Phosphorylated forms of tau are often detected in tau lesions. Measurement of cerebrospinal fluid phospho-tau threonine 181, which can be detected by clone M7004D06, may also have diagnostic utility for several neurological disorders including Alzheimer’s disease.
Phosphorylated forms of tau are often detected in tau lesions. Measurement of cerebrospinal fluid phospho-tau threonine 181, which can be detected by clone M7004D06, may also have diagnostic utility for several neurological disorders including Alzheimer’s disease.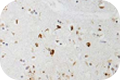 Clone A15091A recognizes phosphorylated tau at residue serine 262. Antibodies against phospho-tau can be used to visualize tau pathologies such as NFTs, neuropil threads, and neuritic plaques by IHC.
Clone A15091A recognizes phosphorylated tau at residue serine 262. Antibodies against phospho-tau can be used to visualize tau pathologies such as NFTs, neuropil threads, and neuritic plaques by IHC.


Follow Us