The most common practice when performing a western blot is to use a secondary antibody to detect the proteins of interest. This is advantageous if there is a need to amplify the signal. However, if the target under study is abundant, and the quality of a directly conjugated primary antibody to Horse Radish Peroxidase (HRP) is high, there is really no advantage or need to use a secondary conjugated antibody. Thus, we are happy to introduce Direct-Blot™, primary antibodies directly conjugated to HRP, to help you speed up, simplify and reduce the cost of your experiment.
Sensitivity
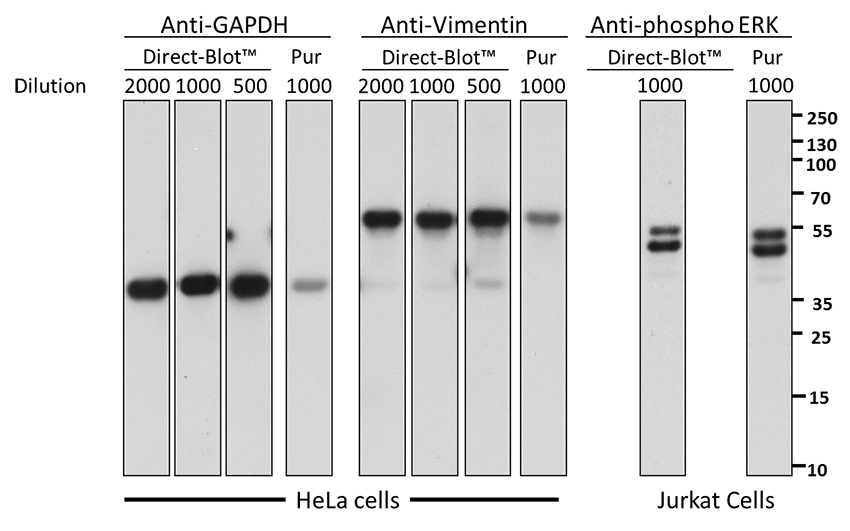
15 µg of total protein from either HeLa cells or Jurkat cells treated with PMA (anti-phospho ERK) was resolved by electrophoresis, transferred to nitrocellulose, and probed with different concentrations of HRP directly conjugated antibodies (Direct-Blot™) or purified monoclonal (Pur) anti-GAPDH (clone FF26A/F9 ), anti-Vimentin (clone O91D3), anti-ERK1/2 Phospho (Thr202/Tyr204) (clone 4B11B69) antibodies at a fixed concentration (0.5 µg/mL). For Direct-Blot™ antibodies, proteins were visualized using chemiluminescence detection directly. For purified antibody, proteins were visualized using a goat anti-mouse-IgG secondary antibody conjugated to HRP (Cat. No. 405306) followed by chemiluminescence detection.
Stability

10 µg of total protein was resolved by electrophoresis and transferred to a PVDF membrane. HRP directly conjugated (Direct-Blot™) antibodies were aliquoted and kept at 4°C, RT and 37° C for 2, 4, 7, and 14 days. Graphs show the percentage of activity of anti-GAPDH and anti-Vimentin at room temperature and 37°C, expressed as percentage of activity at 4°C.
The band intensity quantification was done using the ImageJ software.
 Login/Register
Login/Register 








Follow Us