- Regulatory Status
- RUO
- Other Names
- Natural killer cell stimulatory factor (NKSF), cytotoxic lymphocyte maturation factor (CLMF)
- Ave. Rating
- Submit a Review
- Product Citations
- publications
-
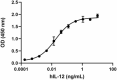
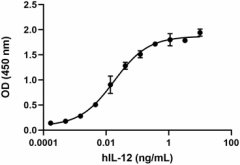
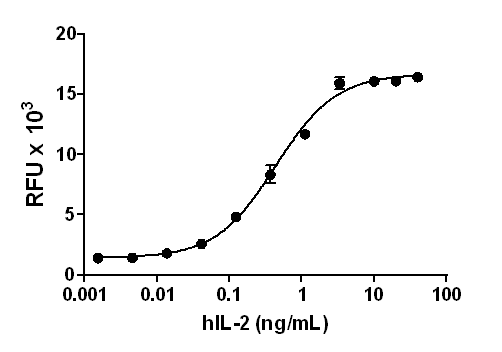
Human IL-12 (p70) induces the production of IFNγ in human activated PBMC in a dose-dependent manner. The ED50 for this effect is 0.01 – 0.1 ng/mL.
IL12 (p70) is a disulfide-linked heterodimer composed of unrelated p40 (glycosylated) and p35 subunits. IL-12 acts as a growth factor for activated human T and NK cells, enhances the lytic activity of human NK cells, and stimulates the production of IFNg by resting human PBMC. IL-12R is formed by two chains, IL-12Rβ1 and IL-12Rβ2. IL-12Rβ1 is associated with the Janus kinase (Jak) Tyk2 and binds IL-12 p40; IL-12Rβ2 is associated with Jak2 and binds either the heterodimer or the p35 chain. Signaling through the IL-12 receptor complex induces phosphorylation, dimerization, and nuclear translocation of several signal transducers and activators of transcription (STAT) family members (STAT1, 3, 4, 5), but most of the biological responses to IL-12 have been attributed to STAT4. IL-12 has been shown to elicit anti-tumor activity in mice and humans. It is believed that the antitumor effects of IL-12 are mediated, at least in part, by indirect mechanisms. Induction of IFN-γ results in the upregulation of class I and class II MHC molecules, adhesion molecules (ICAM-1), nitric oxide production by antigen presenting cells (APC), and the production of additional cytokines, CXCL9 and 10, which in turn mediate angiostatic effects.
Cytokine detection
IL-12, IL-23 and IL-35 share common subunits, utilizing combinations of p40, p19 and p35 proteins. Caution must be used when selecting antibodies and assays when specific identification, measurement, as well as activation state discrimination, is required.
1. Active IL-12 consists of two subunits: p40 + p35.
2. p40 can also exist as a monomer (IL-12 p40) or a homodimer (IL-12 p80).
3. Besides contributing to IL-12, p40 is also found in IL-23, a heterodimer of p40 and p19.
4. Similarly, p35 not only contributes to IL-12, but is also found in the heterodimer IL-35 (p35 and EBI3).
Note that assays using antibodies specific for the p40 subunit will be unable to discriminate between the active IL-12, monomeric p40, dimeric p40, and IL-23; likewise, assays using p35 detection alone will pick up both IL-12 (heterodimer) and IL-35.
Product Details
- Source
- Expressed in insect cells as secreted protein (p35: Accession# NM_000882, p40: Accession# NM_002187)
- Molecular Mass
- The hIL-12 consists of two subunits linked via a disulphide bond: P35 (Accession# NP_000873.2: Arg 57- Ser 253) and P40 (Accession# NP_002178.2: Ile 23-Ser 328). The total predicted molecular weight is 57 kDa. The non-reduced protein migrates at approximately 55 kDa and the DTT-reduced protein produces two bands migrating at approximately 26 kDa and 40 KDa by SDS-PAGE.
- Purity
- >95%, as determined by Coomassie stained SDS-PAGE.
- Formulation
- The protein was 0.22µm filtered in PBS, pH 7.2.
- Endotoxin Level
- Less than 0.01ng per µg cytokine as determined by the LAL method.
- Concentration
- 10 and 25 µg sizes are bottled at 200 µg/mL. 100 µg size and larger sizes are lot-specific and bottled at the concentration indicated on the vial. To obtain lot-specific concentration and expiration, please enter the lot number in our Certificate of Analysis online tool.
- Storage & Handling
- Unopened vial can be stored between 2°C and 8°C for up to 2 weeks, at -20°C for up to six months, or at -70°C or colder until the expiration date. For maximum results, quick spin vial prior to opening. The protein can be aliquoted and stored at -20°C or colder. Stock solutions can also be prepared at 50 - 100 µg/mL in appropriate sterile buffer, carrier protein such as 0.2 - 1% BSA or HSA can be added when preparing the stock solution. Aliquots can be stored between 2°C and 8°C for up to one week and stored at -20°C or colder for up to 3 months. Avoid repeated freeze/thaw cycles.
- Activity
- Human IL-12 (p70) induces the production of IFNγ in human activated PBMC in a dose-dependent manner. The ED50 for this effect is 0.01 – 0.1 ng/mL.
- Application
-
Bioassay
- Application Notes
-
BioLegend carrier-free recombinant proteins provided in liquid format are shipped on blue-ice. Our comparison testing data indicates that when handled and stored as recommended, the liquid format has equal or better stability and shelf-life compared to commercially available lyophilized proteins after reconstitution. Our liquid proteins are verified in-house to maintain activity after shipping on blue ice and are backed by our 100% satisfaction guarantee. If you have any concerns, contact us at tech@biolegend.com.
- Product Citations
-
Antigen Details
- Structure
- Heterodimer
- Distribution
-
IL-12 is produced by monocytes, macrophages, neutrophils, dendritic cells and B cells. In the CNS, astrocytes and microglia are the main sources of IL-12.
- Function
- IL-12 is produced by myeloid cells and DCs in response to microbial stimuli, such as those mediated by bacteria, fungi, viruses, and intracellular parasites. IL-12 drives Th1 differentiation and IFNγ production. IL-12 acts as a bridge between innate resistance and adaptive immunity. IL-12 production by activated APC is suppressed by IL-10. In addition, IL-12 production by macrophages is regulated by TNFα and nitric oxide. TLR-4 and TLR-9 can cooperate to increase selectively IL-12 production by murine dendritic cells.
- Interaction
- Cells of hematopoietic origin express the IL-12R, including NK cells, activated T-cells and dendritic cells.
- Ligand/Receptor
- IL-12 receptor is a heterodimer containing IL-12Rβ1 and IL-12Rβ2 subunits.
- Biology Area
- Immunology, Innate Immunity
- Molecular Family
- Cytokines/Chemokines
- Antigen References
-
1. Schoenhaut DS, et al. 1992. J. Immunol. 148:3433.
2. Manetti R, et al. 1994. J. Exp. Med. 179:1273.
3. Ireland D, et al. 2005. Viral Immunol. 18:397.
4. Moreno SE, et al. 2006. J. Immunol. 177:3218.
5. Lyakh L, et al. 2008. Immunol. Rev. 226:112.
6. Theiner G, et al. 2008. Mol Immunol. 45:244.
7. Zhu S, et al. 2010. J. Immunol. 184:2348. - Gene ID
- 3592 View all products for this Gene ID 3593 View all products for this Gene ID
- UniProt
- View information about IL-12 p70 on UniProt.org
Related Pages & Pathways
Pages
Related FAQs
- Why choose BioLegend recombinant proteins?
-
• Each lot of product is quality-tested for bioactivity as indicated on the data sheet.
• Greater than 95% Purity or higher, tested on every lot of product.
• 100% Satisfaction Guarantee for quality performance, stability, and consistency.
• Ready-to-use liquid format saves time and reduces challenges associated with reconstitution.
• Bulk and customization available. Contact us.
• Learn more about our Recombinant Proteins. - How does the activity of your recombinant proteins compare to competitors?
-
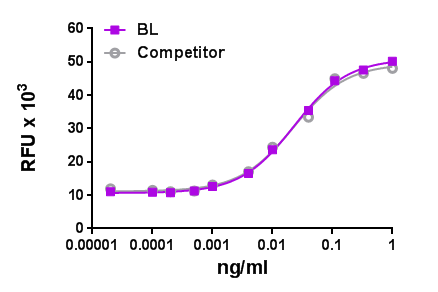
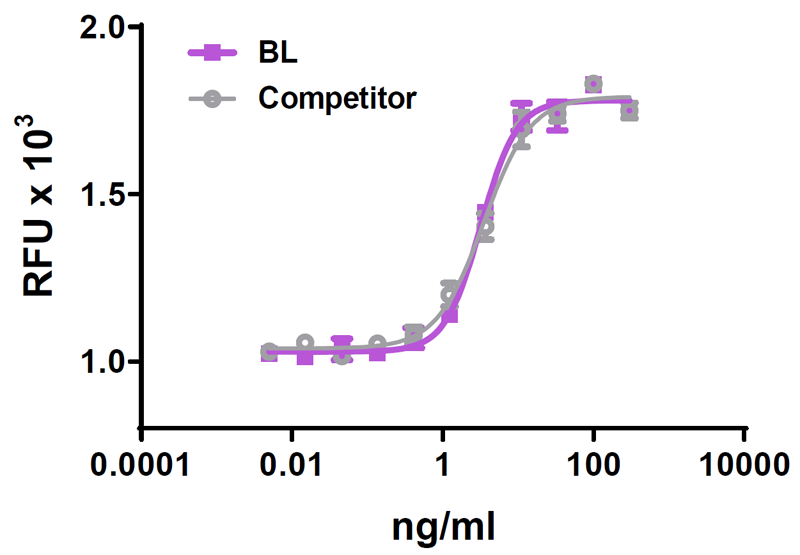
We quality control each and every lot of recombinant protein. Not only do we check its bioactivity, but we also compare it against other commercially available recombinant proteins. We make sure each recombinant protein’s activity is at least as good as or better than the competition’s. In order to provide you with the best possible product, we ensure that our testing process is rigorous and thorough. If you’re curious and eager to make the switch to BioLegend recombinants, contact your sales representative today!
- What is the specific activity or ED50 of my recombinant protein?
-
The specific activity range of the protein is indicated on the product datasheets. Because the exact activity values on a per unit basis can largely fluctuate depending on a number of factors, including the nature of the assay, cell density, age of cells/passage number, culture media used, and end user technique, the specific activity is best defined as a range and we guarantee the specific activity of all our lots will be within the range indicated on the datasheet. Please note this only applies to recombinants labeled for use in bioassays. ELISA standard recombinant proteins are not recommended for bioassay usage as they are not tested for these applications.
- Have your recombinants been tested for stability?
-
Our testing shows that the recombinant proteins are able to withstand room temperature for a week without losing activity. In addition the recombinant proteins were also found to withstand four cycles of freeze and thaw without losing activity.
- Does specific activity of a recombinant protein vary between lots?
-
Specific activity will vary for each lot and for the type of experiment that is done to validate it, but all passed lots will have activity within the established ED50 range for the product and we guarantee that our products will have lot-to-lot consistency. Please conduct an experiment-specific validation to find the optimal ED50 for your system.
- How do you convert activity as an ED50 in ng/ml to a specific activity in Units/mg?
-
Use formula Specific activity (Units/mg) = 10^6/ ED50 (ng/mL)
 Login / Register
Login / Register 









Follow Us