As cell therapies continue to develop and expand, biomanufacturers understand that high-quality raw materials play an important, early role in controlling the overall quality, safety, and efficacy of the final products. Our expanding line of GMP recombinant proteins are intended for research or ex vivo cell bioprocessing. These proteins are manufactured and tested in a dedicated GMP facility that is ISO 13485:2016 and MDSAP certified, monitored through independent QA oversight. They are manufactured and tested in accordance with USP Chapter 1043, Ancillary Materials for Cell, Gene and Tissue-Engineered Products and Ph. Eur. Chapter 5.2.12 and come with complete raw material traceability and documentation support.
Find complete regulatory documentations here.
Our GMP recombinant proteins are ideal for use in expansion, polarization, and differentiation of cells for research and ex vivo cell processing applications. BSA or other equivalent carrier proteins are typically added for protein stability and to increase shelf life. However, addition of carrier proteins may produce nonrelated or undesired effects on experiments. Our GMP recombinant proteins are carrier-free, thus eliminating this risk.
View all GMP recombinant proteins.
Contact our sales team for help with custom products or bulk requests.
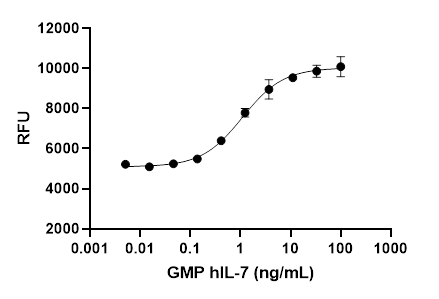
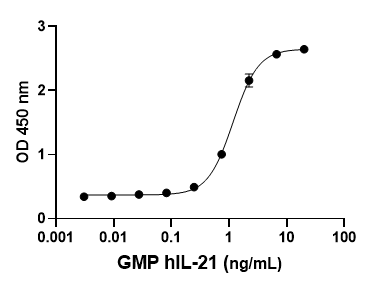
- Each lot of our GMP recombinant proteins undergoes extensive QC testing, including:
- Biological Activity Validation
- Purity Testing
- Concentration Check
- Endotoxin Testing (<0.1 EU/µg)
- Bioburden Testing
 Login / Register
Login / Register 







Follow Us