- Regulatory Status
- RUO
- Other Names
- Calcein, Calcein AM
- Ave. Rating
- Submit a Review
- Product Citations
- publications
-

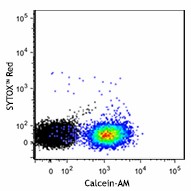
Fresh (top) or day-old C57BL/6 splenocytes (bottom) were stained with Calcein-AM and a cell-impermeant nucleic acid dye, SYTOX™ Red (colored). Black figure represents unstained splenocytes. -

| Cat # | Size | Price | Quantity Check Availability | Save | ||
|---|---|---|---|---|---|---|
| 425201 | 10 x 50 µg | 132€ | ||||
Calcein-AM is a fluorogenic, cell-permeant fluorescent probe that indicates cellular health. When the acetoxymethyl ester is intact, this probe is nonfluorescent until acted upon by nonspecific esterases present within the healthy, live cells. Cleaving the AM ester allows the probe to excite and emit at 488nm/ 520nm respectively. The signal of Calcein-AM is proportional to cell vitality, as esterase activity decreases in cells with poor vitality. This cell-permeant probe is not retained with fixation and peameabilization.
Product DetailsProduct Details
- Preparation
- Calcein-AM labeling kit consists of lyophilized Calcein-AM and anhydrous DMSO. For reconstitution, bring the kit to room temperature and add 50 µL of DMSO to one vial of Calcein-AM dye until fully dissolved.
- Storage & Handling
- Upon receipt, Calcein-AM should be stored at -20°C. Do not open vials until needed. Once the DMSO is added to the Calcein-AM, use immediately or store at -20°C in a dry place and protected from light, preferably in a desiccator or in a container with desiccant for no more than one month.
- Application
-
FC - Quality tested
- Recommended Usage
-
This lot has been tested by flow cytometric analysis of cell vitality. It can be used at concentrations ranging from 0.1 – 0.001 µM for cell labeling. It is recommended that the reagent be titrated for optimal performance for each cell type, culturing condition, or application.
- Application Notes
-
The molecular weight of Calcein-AM is 994 Da. The maximum excitation and emission wavelengths of Calcein-AM are 488 nm/520 nm, respectively. Each 50 µg of Calcein-AM may be reconstituted with 50 µL of anhydrous DMSO to yield a stock concentration of 1 mM.
Materials Provided:
10 vials x 50 µg Calcein-AM
2 vials of 500 µl anhydrous DMSO
Calcein-AM Labeling Procedure:
1. Prior to reconstitution, spin down the vial of lyophilized reagent in a microcentrofuge to ensure the reagent is at the bottom of the vial.
2. Prepare a 1 mM stock solution by reconstituting 1 vial of lyophilized Calcein-AM dye with 50 µl of anhydrous DMSO.
3. Prepare a 1 µM working solution by diluting 1 µL of 1 mM Calcein-AM stock solution in 1 mL PBS.
4. Spin down cells and adjust the cell suspension to 1x107 cells/mL in PBS.
5. Add 10µl of the 1 µM Calcein-AM working solution to each mL of cell suspension for a final concentration of 0.01 µM.
6. Incubate cells for 20 minutes at room temperature or at 37°C and keep protected from light.
7. Pellet cells and resuspend in pre-warmed cell culture medium.
8. Incubate cells for 10 minutes to ensure optimal retention of the Calcein-AM.
9. After incubation, Calcein-AM labeled cells are ready for downstream applications or analysis. - Application References
-
- Mariappan MR, et al. 1999. IEEE Eng. Med. Biol. Mag. 18:22.
- Mironov SL, et al. 2005. J. Biol. Chem. 280:715.
- Tian F, et al. 2009. Blood. 113:5352.
- Ansbro MR, et al. 2013. PLoS One 8(4):e60334. (IF)
- Gonzalez JM, et al. 2013. Invest. Ophthalmol. Vis. Sci. 54:1039. (IF)
- Godin LM, et al. 2011. Am. J. Physiol. Lung Cell Mol. Physiol. 300:L615. (IF)
- Product Citations
-
Antigen Details
- Structure
- Acetoxymethyl ester.
- Distribution
-
Cytoplasmic.
- Function
- Cell vitality indicator.
- Interaction
- Nonspecific esterases.
- Biology Area
- Cell Proliferation and Viability
- Gene ID
- NA
 Login / Register
Login / Register 













Follow Us