- Regulatory Status
- RUO
- Other Names
- Bone morphogenetic protein 2 (BMP-2), bone morphogenetic protein 2A (BMP-2A), BDA2
- Ave. Rating
- Submit a Review
- Product Citations
- publications
-
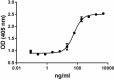
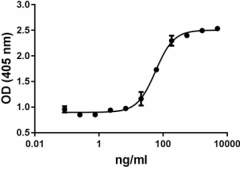
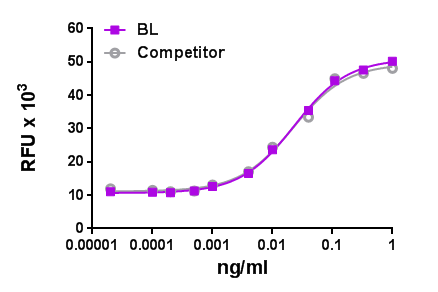
BMP-2 induces ALP production in mouse chrondrogenic cell line ATDC5. -
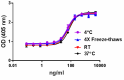
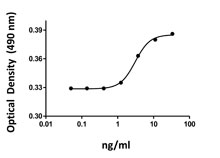
Stability test for Human/Mouse/Rat BMP-2:BMP-2 was aliquoted in 4 mM HCl at 0.1 mg/ml. The aliquots were treated as follows: at 4°C (control), room temperature (RT), and 37°C for one week, and one aliquot was freeze/thawed four times (4x free-thaws). After this procedure, the samples were tested for their ability to induce alkaline phosphatase in the mouse chrondrogenic cell line ATDC5.
Bone morphogenetic protein 2 (BMP-2) belongs to the TGF-β superfamily. Like other members of BMP family, BMP-2 is synthesized as an inactive propeptide precursor which dimerizes and then it is further processed into mature form by proprotein convertases (PCs). Some evidence indicated that PC5/6A and Factor VII-activating protease (FSAP) are involved in maturation of BMP-2. Mature BMP-2 is a 26 kD protein composed of 114 amino acids, forming three intramolecular and one intermolecular disulfide bond. BMP-2 forms homodimer or heterodimer with other BMP proteins, including BMP-4, BMP-6 and BMP-7. BMP-2 signal through heterodimeric serine/threonine kinase receptors composed of a type I (BMPR1a/ALK3, BMPR1b/ALK6, ActRIa/ALK2) and a type II (BMPR2, ACVR2a/ActRIIA, ACVR2b/ActRIIB). BMP-2 binds to the type I receptor with high affinity, in turn recruits the type II receptor. BMP-2 stimulation initiates receptor shutting, leading to receptor clustering and activation of the downstream signaling. BMP-2 signals via canonical Smad or other downstream kinase, such as p38 and JNK in a context-dependent manner. BMP-2 is involved in several processes, including cartilage and bone formation, differentiation, and embryogenesis. BMP-2 induces osteogenic differentiation in human mesenchymal stem cells and myogenic cells. BMP-2 induces cartilage repair and remodeling by stimulating chondrocyte proliferation and expression of matrix proteins. BMP-2/BMP-7 heterodimer is more potent in the induction of bone formation in vivo than BMP-2 homodimer. BMP-2-deficiency leads to embryonic lethality with abnormal cardiac development, malformation of the amnion/chorion, severe chondrodysplasia, and defects in myocardial patterning, suggesting that BMP-2 mediates organ morphogenesis. Noggin is an antagonist to reverse BMP-2-mediated effect and its expression is induced by BMP-2 in osteoblasts as a negative feedback loop. In addition, BMP-2 stimulates epithelial to mesenchymal cell transformation through TGF-β type III receptor.
Product DetailsProduct Details
- Source
- Human/Mouse/Rat BMP-2, amino acid Ala284-Arg396 (Accession# P12643), was expressed in E. coli.
- Molecular Mass
- The 113 amino acid recombinant protein has a predicted molecular mass of approximately 12.8 kD. The DTT-reduced and non-reduced protein migrates at approximately 13 and 26 kD respectively by SDS-PAGE. The predicted N-terminal amino acid is Ala.
- Purity
- > 98%, as determined by Coomassie stained SDS-PAGE.
- Formulation
- Sterile-filtered with 0.2 µm filter, solution comprised of 4 mM HCl.
- Endotoxin Level
- Less than 0.1 EU/µg cytokine as determined by the LAL method.
- Concentration
- 10-25 µg sizes are bottled at 200 µg/mL. 100 µg and larger sizes and larger are bottled at the concentration indicated on the vial.
- Storage & Handling
- Unopened vial can be stored between 2°C and 8°C for up to 2 weeks, at -20°C for up to six months, or at -70°C or colder until the expiration date. For maximum results, quick spin vial prior to opening. The protein can be aliquoted and stored at -20°C or colder. Stock solutions can also be prepared at 50 - 100 µg/mL in appropriate sterile buffer, carrier protein such as 0.2 - 1% BSA or HSA can be added when preparing the stock solution. Aliquots can be stored between 2°C and 8°C for up to one week and stored at -20°C or colder for up to 3 months. Avoid repeated freeze/thaw cycles.
- Activity
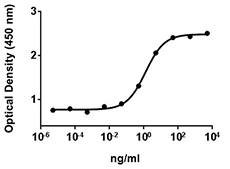
- The ED50= 40-200 ng/ml, as determined by induction of alkaline phosphatase (ALP) production in the mouse chrondrogenic cell line ATDC5 in a dose dependent manner.
- Application
-
Bioassay
- Application Notes
-
BioLegend carrier-free recombinant proteins provided in liquid format are shipped on blue-ice. Our comparison testing data indicates that when handled and stored as recommended, the liquid format has equal or better stability and shelf-life compared to commercially available lyophilized proteins after reconstitution. Our liquid proteins are verified in-house to maintain activity after shipping on blue ice and are backed by our 100% satisfaction guarantee. If you have any concerns, contact us at tech@biolegend.com.
Antigen Details
- Structure
- Disulfide-linked homodimer
- Distribution
-
Abundant in lung, spleen and colon in human, low expression but detectable in heart, kidney, brain, liver, skeletal muscle, pancreases, placenta, prostate, ovary, and small intestine.
- Function
- BMP-2 is involved in osteogenesis, cartilage repair, and organogenesis.
- Interaction
- Chondroblast, osteoblast, proprotein convertases, BMP family members
- Ligand/Receptor
- BMP receptor type IA (BMPR1a/ALK3), type IB (BMPR1b/ALK6), Activin receptor type IA (ACVR1a/ActRIa/ALK2) and BMP receptor type II (BMPR2), Activin receptors type IIA (ACVR2a/ActRIIA), type IIB (ACVR2b/ActRIIB)
- Bioactivity
- BMP-2 induces alkaline phosphatase in ATDC5 mouse chondrogenic cells.
- Cell Type
- Embryonic Stem Cells
- Biology Area
- Angiogenesis, Cell Biology, Neuroscience, Stem Cells, Synaptic Biology
- Molecular Family
- Cytokines/Chemokines, Growth Factors
- Antigen References
-
- Bragdon B, et al. 2011. Cell Signal 23: 609.
- Israel DI, et al. 1992. Growth Factor. 7: 139.
- Lee SN, et al. 2015. Am. J. Respir. Cell Mol. Biol. 52: 749.
- Roedel EK, et al. 2013. J. Biol. Chem. 288, 7193.
- Nohe A, et al. 2002. J. Biol. Chem. 277: 5330.
- Zhou AJ, et al. 2012. Growth Factors 30: 267.
- Miyazono K, et al. 2010. J. Biochem. 147: 35.
- Nohe A et al. 2004. J. Cell Sci. 118: 643.
- Hay E, et al. 2001. J. Biol. Chem. 276: 29028.
- Wozney JM, et al. 1988. Science 242: 1528.
- Ryoo HM, et al. 2006. Gene 366: 51.
- Lavery K, et al. 2008. J. Biol. Chem. 283: 20948.
- Israel DI, et al. 1996. Growth Factors 13: 291.
- Mundy G, et al. 1999. Science 286: 1946.
- De Luca F et al. 2001. Endocrinology 142: 430.
- Blaney Davidson EN, et al. 2007. Arthritis Res. Ther. 9: R102.
- Zhang H, Bradley A. 1996. Development 122: 2977.
- Pera MF, et al. 2004. J. Cell Sci. 117: 1269.
- Wang RN, et al. 2014. Genes Dis. 87-105.
- Kirkbride KC, et al. 2008. J. Biol. Chem. 283: 7628.
- Gene ID
- 650 View all products for this Gene ID
- UniProt
- View information about BMP-2 on UniProt.org
Related FAQs
- Why choose BioLegend recombinant proteins?
-
• Each lot of product is quality-tested for bioactivity as indicated on the data sheet.
• Greater than 95% Purity or higher, tested on every lot of product.
• 100% Satisfaction Guarantee for quality performance, stability, and consistency.
• Ready-to-use liquid format saves time and reduces challenges associated with reconstitution.
• Bulk and customization available. Contact us.
• Learn more about our Recombinant Proteins. - How does the activity of your recombinant proteins compare to competitors?
-
We quality control each and every lot of recombinant protein. Not only do we check its bioactivity, but we also compare it against other commercially available recombinant proteins. We make sure each recombinant protein’s activity is at least as good as or better than the competition’s. In order to provide you with the best possible product, we ensure that our testing process is rigorous and thorough. If you’re curious and eager to make the switch to BioLegend recombinants, contact your sales representative today!
- What is the specific activity or ED50 of my recombinant protein?
-
The specific activity range of the protein is indicated on the product datasheets. Because the exact activity values on a per unit basis can largely fluctuate depending on a number of factors, including the nature of the assay, cell density, age of cells/passage number, culture media used, and end user technique, the specific activity is best defined as a range and we guarantee the specific activity of all our lots will be within the range indicated on the datasheet. Please note this only applies to recombinants labeled for use in bioassays. ELISA standard recombinant proteins are not recommended for bioassay usage as they are not tested for these applications.
- Have your recombinants been tested for stability?
-
Our testing shows that the recombinant proteins are able to withstand room temperature for a week without losing activity. In addition the recombinant proteins were also found to withstand four cycles of freeze and thaw without losing activity.
- Does specific activity of a recombinant protein vary between lots?
-
Specific activity will vary for each lot and for the type of experiment that is done to validate it, but all passed lots will have activity within the established ED50 range for the product and we guarantee that our products will have lot-to-lot consistency. Please conduct an experiment-specific validation to find the optimal ED50 for your system.
- How do you convert activity as an ED50 in ng/ml to a specific activity in Units/mg?
-
Use formula Specific activity (Units/mg) = 10^6/ ED50 (ng/mL)
 Login / Register
Login / Register 








Follow Us