- Clone
- Poly19055 (See other available formats)
- Regulatory Status
- IVD
- Isotype
- Rabbit Polyclonal
-
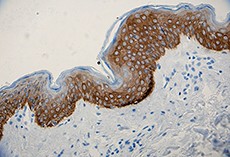
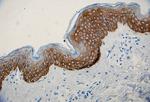
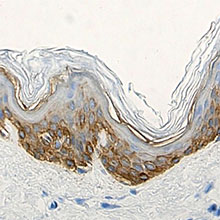
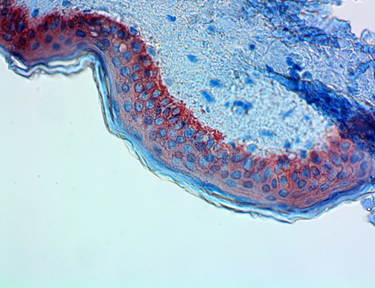
Anti-K5 antibody (clone Poly19055) showing strong cytoplasmic staining of the basal cells of normal human skin epidermis. -

-
| Cat # | Size | Price | Quantity Check Availability | Save | ||
|---|---|---|---|---|---|---|
| 905501 | 100 µL | 352€ | ||||
Product Details
- Product Information
-
Intended Use:
In Vitro Diagnostic (IVD). Use in immunohistochemistry (IHC) test methods only.
The polyclonal antibody Poly19055 is used for the in vitro examination of frozen or paraffin-embedded human skin tissue sections using immunohistochemistry (IHC) methods for the qualitative identification of Keratin 5. The clinical interpretation of any staining or its absence should be complemented by morphological studies and proper controls and should be evaluated within the context of the patient's clinical history and other diagnostic tests by a qualified pathologist.
- Reactivity
- Human
- Immunogen
- This polyclonal antibody was raised against a peptide sequence derived from the C-terminus of the mouse keratin 5 protein.
- Formulation
- Phosphate-buffered solution + 0.03% Thimerosal.
- Preparation
- The antibody was purified by peptide affinity chromatography.
- Concentration
- 1.0 mg/mL
- Storage & Handling
- When stored at ≤ -20°C, product is stable until the date shown on the label. Avoid repeated freeze-thaw cycles to prevent denaturing the antibody. If thawed and stored at 2-8°C, product is stable for 24 months from the date of thaw or until the expiry date on the label, whichever comes first.
- Recommended Usage
-
Each lot of this antibody is quality control tested by immunohistochemical staining of formalin-fixed paraffin-embedded sections of normal human skin. Frozen skin has been verified during product development.
The optimal working dilution should be determined for each specific assay condition.
• IHC: 1:2,000-4,000 with either biotin based detection systems such as USA Ultra Streptavidin Detection (Cat. No. 929501).
Tissue Sections: Paraffin-embedded tissues, frozen tissues
Pretreatment: For optimal staining, the sections should be pretreated with an antigen unmasking solution such as Citrate Buffer Retrieval solution (Cat. No. 928502).
Incubation: 60 minutes at room temperature - Application References
-
- Hübner A, et al. JNK and PTEN cooperatively control the development of invasive adenocarcinoma of the prostate. PNAS, 109: 12046-12051, Jul 2012. [IHC] PubMed
- Kierszenbaum AL, Rivkin E, Tres LL. Acroplaxome, an F-actin-keratin-containing plate, anchors the acrosome to the nucleus during shaping of the spermatid head. Mol Biol Cell 14(11):4628-40, 2003. [IHC] PubMed
- Hu Y, Baud V, Oga T, Kim KI, Yoshida K, Karin M. IKKalpha controls formation of the epidermis independently of NF-kappaB. Nature 410:710-714, 2001.
- Yuspa SH, Kilkenny AD, Steinert PM, Roop DR. Expression of murine epidermal differentiation markers is tightly regulated by restricted extracellular calcium concentrations in-vitro. J Cell Biol 109:1207-1217, 1989.
- Roop DR, Cheng CK, Titterington L, Meyers CA, Stanley JR, Steinert PM, Yuspa SH. Synthetic peptides corresponding to keratin subunits elicit highly specific antibodies. J Biol Chem 259:8037-8040, 1984.
- Liang Y, 2011 Patholog Res Int. 2011:936794. (IHC) PubMed
- Easter SL, et al. 2014. PLoS One. 9:113247. (IF, WB) PubMed
- Osmanagic-Myers S, et al.2006. J Cell Biol. 174:557. (WB) PubMed
- Kittrell FS, et al. 2011. Breast Cancer Res.13:41. (IF) PubMed
- Croyle MJ, et al. Role of epidermal primary cilia in the homeostasis of skin and hair follicles. Development, 138: 1675-1685, May 2011. [IHC, IF] PubMed
- Bayo P, Sanchis A, Bravo A, Cascallana JL, Buder K, Tuckermann J, Schütz G, and PTrez P. Glucocorticoid Receptor Is Required for Skin Barrier Competence. Endocrinology, 149: 1377-1388, Mar 2008. [IHC] PubMed
- RRID
-
AB_2565050 (BioLegend Cat. No. 905501)
- Disclaimer
-
WARNINGS AND PRECAUTIONS
- Use appropriate personal protective equipment and safety practices per universal precautions when working with this reagent. Refer to the reagent safety data sheet.
- All specimens, samples and any material coming in contact with them should be considered potentially infectious and should be disposed of with proper precautions and in accordance with federal, state and local regulations.
- Do not use this reagent beyond the expiration date stated on the label.
- Do not use this reagent if it appears cloudy or if there is any change in the appearance of the reagent as these may be indication of possible deterioration.
Customers Also Purchased
Compare Data Across All Formats
This data display is provided for general comparisons between formats.
Your actual data may vary due to variations in samples, target cells, instruments and their settings, staining conditions, and other factors.
If you need assistance with selecting the best format contact our expert technical support team.
-
Keratin 5 Polyclonal Antibody, Purified
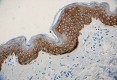
Anti-K5 antibody (clone Poly19055) showing strong cytoplasmi... 

-
Purified anti-Keratin 5
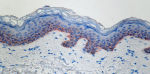
IHC staining of anti-Keratin 5 antibody (Poly19055) on forma... 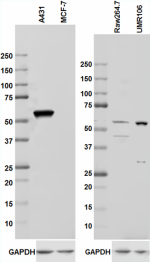
Total lysates (15 µg protein) from A431 (Human, Positive con... 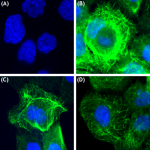
Immunofluorescence of A431 cells with (A) Rabbit Isotype con...
 Login / Register
Login / Register 







Follow Us