- Regulatory Status
- RUO
- Other Names
- Macrophage metalloelastase (MME), Matrix metalloproteinase 12
- Ave. Rating
- Submit a Review
- Product Citations
- publications
-
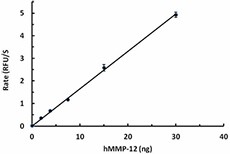
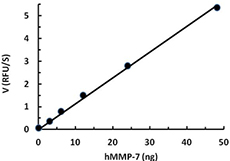
hMMP-12 (50 µg/mL) was activated in the presence of 1 mM APMA for 1 hr at 37 °C. The activity of hMMP-12 was measured with 10 µM of fluorogenic MMP substrate, Mca-PLGL-Dpa-AR-NH2, in the presence of 1.88, 3.75, 7.5, 15, 30 ng of activated hMMP-12. The activity of activated hMMP-12 is greater than 750 pmole/min/µg.
| Cat # | Size | Price | Quantity Check Availability | Save | ||
|---|---|---|---|---|---|---|
| 764202 | 10 µg | 179€ | ||||
| 764204 | 25 µg | 329€ | ||||
Matrix metalloproteinases (MMPs) are a family of zinc-dependent endopeptidases that degrade components of the extracellular matrix (ECM) and play essential roles in various physiological and pathological processes such as morphogenesis, differentiation, angiogenesis, tissue remodeling, and tumor invasion. A typical MMP consists of a propeptide of about 80 amino acids, a catalytic metalloproteinase domain of about 170 amino acids, a linker peptide of variable lengths and a hemopexin domain of about 200 amino acids. The zinc binding motif HEXXHXXGXXH in the catalytic domain, and the ‘‘cysteine switch’’ motif PRCGXPD in the propeptide are common structural signatures. MMP-12 (macrophage elastase) is a 54 kDa proenzyme consisted of propeptide, catalytic metalloproteinase domain, and hemopexin domain. The predicted human 54-kD protein is processed by loss of both N- and C-terminal residues to a 22-kD mature form. It is a major proteinase that degrades elastin. MMP-12 can also degrade a broad spectrum of substrates, including type IV collagen, fibronectin, laminin, vitronectin, proteoglycans, chondroitin sulfate, myelin basic protein, alpha 1-antitrypsin, and plasminogen. MMP-12 is mainly produced by macrophages and has been shown to be associated with inflammatory skin diseases, atherosclerosis, aneurysms, and cancers. MMP12 can exhibit both proinflammatory and anti-inflammatory activity in a tissue- or disease context-dependent manner. The purified recombinant MMP-12 is consisted of pro-peptide and catalytic domain.
Product DetailsProduct Details
- Source
- Human MMP12, amino acids Leu17-Gly263 (Pro-peptide and catalytic domain; Accession #NM_006287) with a C-terminal TG-8H-GGQ tag was expressed in CHO cells.
- Molecular Mass
- The 260 amino acid recombinant protein has a predicted molecular mass of approximately 29.2 kDa. The non-reduced and DTT-reduced proteins migrate at 32 - 35 kDa by SDS-PAGE.
- Purity
- > 90%, as determined by Coomasie stained SDS-PAGE
- Formulation
- 0.22 µm filtered protein solution is in 10 mM MES, 5 mM CaCl2, 150 mM NaCl and 50% glycerol at pH 5.5.
- Endotoxin Level
- Less than 1.0 EU per µg of protein as determined by the LAL method
- Concentration
- 10 and 25 µg sizes are bottled at 200 µg/mL. 100 µg size and larger sizes are lot-specific and bottled at the concentration indicated on the vial. To obtain lot-specific concentration and expiration, please enter the lot number in our Certificate of Analysis online tool.
- Storage & Handling
- Unopened vial can be stored at -20 °C or -70°C for six months. For maximum results, quick spin vial prior to opening. Avoid repeated freeze/thaw cycles.
- Activity
- Human MMP-7 cleaves a fluorogenic peptide substrate Mca-PLGL(Dpa)AR with a specific activity value > 750 pmol/µg/min
- Application
-
Bioassay
- Application Notes
-
Human MMP-12 Activity assay
Human MMP-12 (hMMP-12) activity is measured by its ability to cleave a fluorogenic peptide substrate Mca-PLGL(Dpa)AR after its activation in the presence of p-Aminophenylmercuric acid. The increase of the product is monitored by increase in intensity of fluorescence at 405 nm with excitation at 320 nm. This protein is in the latent form and needs to be activated for bioassay.
Materials
- Assay Buffer: TCNB (50 mM Tris, 10 mM CaCl2, 150 mM NaCl, 0.05% Brij-35, pH 7.5)
- Recombinant Human MMP-12
- MCa-PLGL(Dpa)AR (AnaSpec Cat # AS27076; 2 mM in DMSO)
- P-Aminophenylmercuric acid (Calbiochem Cat# 164610; 100 mM in DMSO)
Activity assay procedures
- Dilute hMMP-12 in the assay buffer at 100 µg/mL.
- Activate hMMP-12 by adding APMA to a final concentration of 1 mM.
- Incubate the activating mixture for 1 hr at 37 °C.
- Dilute the activated hMMP-12 to 0.4 ug/mL (0.4 ng/µL) in Assay Buffer.
- Dilute the substrate at 20 µM in Assay Buffer.
- Load into a well plate 50 μL of the 0.4 ng/μL hMM-12 and start the reaction by adding 50 μL of 20 μM Substrate. Include a substrate blank containing 50 μL Assay Buffer and 50 μL of 20 μM Substrate without any hMMP-12.
- Read the product formation by measuring 320/ 405 nm (Excitation/Emission) in kinetic mode for 5 minutes.
The final hMMP-12 concentration is 0.2 µg/mL (0.2 ng/µl, 20 ng)
The final concentration of the substrate is 10 µM.
BioLegend carrier-free recombinant proteins provided in liquid format are shipped on blue-ice. Our comparison testing data indicates that when handled and stored as recommended, the liquid format has equal or better stability and shelf-life compared to commercially available lyophilized proteins after reconstitution. Our liquid proteins are verified in-house to maintain activity after shipping on blue ice and are backed by our 100% satisfaction guarantee. If you have any concerns, contact us at tech@biolegend.com.
Antigen Details
- Structure
- Monomer
- Distribution
-
High expression in macrophage; Moderate expression in Fibroblast cell
- Function
- Wound healing; Extracellular matrix diassembly
- Interaction
- Extracellular matrix
- Ligand/Receptor
- TIMP-I, TIMP-2, TIMP-4
- Bioactivity
- Regulate blood coagulation by inhibit FXa and FVIIa
- Biology Area
- Angiogenesis, Cell Adhesion, Cell Biology, Neuroinflammation, Neuroscience, Stem Cells
- Molecular Family
- Enzymes and Regulators
- Antigen References
-
1. Liu S-L et al. 2015. Sci. Rep. 5:17189.
2. Laurenzana A et al. 2014. Oncotarget. 5:3711.
3. Bellac CL et al. 2014. Cell. Rep. 9:618.
4. Lee JT et al. 2014. Endocrinology. 155:3409.
5. Tallant C et al. 2010. Biochem. Biophys. Acta. 1803:20.
6. Nagase H, et al. 2006. Cardiovasc. Res. 69(3):562-73.
7. Rundhaug JE, 2005. J. Cell. Mol. Med. 9:267 - Gene ID
- 4321 View all products for this Gene ID
- UniProt
- View information about MMP-12 on UniProt.org
Related FAQs
- Why choose BioLegend recombinant proteins?
-
• Each lot of product is quality-tested for bioactivity as indicated on the data sheet.
• Greater than 95% Purity or higher, tested on every lot of product.
• 100% Satisfaction Guarantee for quality performance, stability, and consistency.
• Ready-to-use liquid format saves time and reduces challenges associated with reconstitution.
• Bulk and customization available. Contact us.
• Learn more about our Recombinant Proteins. - How does the activity of your recombinant proteins compare to competitors?
-
We quality control each and every lot of recombinant protein. Not only do we check its bioactivity, but we also compare it against other commercially available recombinant proteins. We make sure each recombinant protein’s activity is at least as good as or better than the competition’s. In order to provide you with the best possible product, we ensure that our testing process is rigorous and thorough. If you’re curious and eager to make the switch to BioLegend recombinants, contact your sales representative today!
- What is the specific activity or ED50 of my recombinant protein?
-
The specific activity range of the protein is indicated on the product datasheets. Because the exact activity values on a per unit basis can largely fluctuate depending on a number of factors, including the nature of the assay, cell density, age of cells/passage number, culture media used, and end user technique, the specific activity is best defined as a range and we guarantee the specific activity of all our lots will be within the range indicated on the datasheet. Please note this only applies to recombinants labeled for use in bioassays. ELISA standard recombinant proteins are not recommended for bioassay usage as they are not tested for these applications.
- Have your recombinants been tested for stability?
-
Our testing shows that the recombinant proteins are able to withstand room temperature for a week without losing activity. In addition the recombinant proteins were also found to withstand four cycles of freeze and thaw without losing activity.
- Does specific activity of a recombinant protein vary between lots?
-
Specific activity will vary for each lot and for the type of experiment that is done to validate it, but all passed lots will have activity within the established ED50 range for the product and we guarantee that our products will have lot-to-lot consistency. Please conduct an experiment-specific validation to find the optimal ED50 for your system.
- How do you convert activity as an ED50 in ng/ml to a specific activity in Units/mg?
-
Use formula Specific activity (Units/mg) = 10^6/ ED50 (ng/mL)
 Login / Register
Login / Register 








Follow Us