- Regulatory Status
- RUO
- Other Names
- CE, CATE
- Ave. Rating
- Submit a Review
- Product Citations
- publications
-
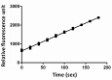
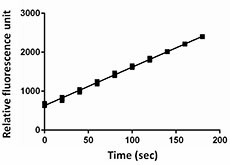
CTSE enzyme activity was measured using a fluorogenic peptide substrate, Mca-Pro-Leu-Gly-Leu-Dpa-Ala-Arg-NH2.
| Cat # | Size | Price | Quantity Check Availability | Save | ||
|---|---|---|---|---|---|---|
| 599302 | 10 µg | $229 | ||||
Cathepesin E (CTSE) is an intracellular aspartic protease that was originally identified as a cathepsin D-like acid protease. CTSE and CTSD have similar substrate specificities and CTSE is active in acidic conditions in a pH range from 2.5 to 5.5. In vitro experiments have identified several CTSE substrates including insulin beta chain, neurokinin, and FGF. Although the function of CTSE is not completely understood, it has been implicated in several physiological and pathological processes. CTSE is required for antigen presentation on class II MHC molecules and subsequently, CTSE-deficient mice have increased susceptibility to bacterial infections. CTSE-deficient macrophages also show abnormalities, such as autophagy. Like many other cathepsins, CTSE has emerged as a therapy target for cancers, such as pancreatic ductal adenocarcinoma (PDAC). In addition to PDAC, CTSE is also overexpressed in gastric carcinomas and cervical and lung adenocarcinomas. The possible involvement of CTSE in neurodegeneration has also been reported.
Product DetailsProduct Details
- Source
- Human Cathepsin E, amino acids Gln18 - Pro396 (Accession# NP_001901) with a C-terminal 10-His tag, was expressed in 293E cells.
- Molecular Mass
- The 389 amino acid recombinant protein has a predicted molecular mass of approximately 42.3 kD. The DTT-reduced protein migrates at approximately 45 kD and the non-reduced protein migrates at approximately 90 kD by SDS-PAGE. The N-terminal amino acid is Gln.
- Purity
- >95%, as determined by Coomassie stained SDS-PAGE.
- Formulation
- 0.22 µm filtered protein solution is in 20 mM MES, 150 mM NaCl, pH 6.5.
- Endotoxin Level
- Less than 0.01 ng per µg cytokine as determined by the LAL method.
- Concentration
- 10 and 25 µg sizes are bottled at 100 µg/mL.
- Storage & Handling
- Unopened vial can be stored at -20°C for one month, or at -70°C for six months. For maximum results, quick spin vial prior to opening. Avoid repeated freeze/thaw cycles.
- Activity
- After auto-activation, CTSE activity is determined by its ability to cleave the fluorogenic peptide substrate, Mca-Pro-Leu-Gly-Leu-Dpa-Ala-Arg-NH2. The specific activity is >1,500 pmol/min/µg.
- Application
-
Bioassay
- Application Notes
-
Activation Protocol
Step 1: Dilute recombinant human CTSE to 1 μg/ml in assay buffer (0.1 M NaOAc, 0.5 M NaCl, pH 3.5) and incubate the protein at 25°C for 30 min to activate CTSE. After activation, dilute activated CTSE to 0.2 μg/ml using assay buffer.
Step 2: Dilute fluorogenic peptide substrate (Mca-Pro-Leu-Gly-Leu-Dpa-Ala-Arg-NH2) to 40 μM using assay buffer.
Step 3: Perform the assay in a 96-well, black, flat-bottomed plate. Add 50 μl of the diluted, activated CTSE (0.2 μg/ml) to a black well and start the reaction by adding 50 μl of 40 nm substrate. Assay is read on a fluorometer at excitation and emission wavelengths of 320 nm and 405 nm (top read).
Note: This protein is in the latent form and needs to be activated for bioassay.
BioLegend carrier-free recombinant proteins provided in liquid format are shipped on blue-ice. Our comparison testing data indicates that when handled and stored as recommended, the liquid format has equal or better stability and shelf-life compared to commercially available lyophilized proteins after reconstitution. Our liquid proteins are verified in-house to maintain activity after shipping on blue ice and are backed by our 100% satisfaction guarantee. If you have any concerns, contact us at tech@biolegend.com.
Antigen Details
- Structure
- Disulfide-linked dimer.
- Distribution
-
CTSE is mainly expressed by immune cells including macrophages, lymphocytes, microglia, dendritic cells, and activated phagocytes. The intracellular localization of CTSE is cell type dependent. The mature form of CTSE is localized mainly in endosomal compartments in antigen-presenting cells; the inactive pro-form exists in the plasma membrane, Golgi apparatus, and in other tissues and cells.
- Function
- CTSE expression is negatively regulated by the MHC class II transactivator.
- Ligand/Receptor
- Substrate P, neurokinin A, eledoisin, kassinin, FGF, b-endorphin, porcine renin substrate.
- Biology Area
- Apoptosis/Tumor Suppressors/Cell Death, Cell Biology, Neurodegeneration, Neuroscience, Neuroscience Cell Markers, Protein Trafficking and Clearance, Stem Cells
- Molecular Family
- Enzymes and Regulators, Lysosomal Markers
- Antigen References
-
1. Nishioku T, et al. 2002. J. Biol. Chem. 277:4816.
2. Sastradipura D, et al. 1998. J. Neurochem. 70: 2045.
3. Presta M, et al. 2005. Cytokine Growth Factor Rev. 16:159.
4. Zakrzewska M, et al. 2008. Crit. Rev. Clin. Lab. Sci. 45:91.
5. Saku T, et al. 1991. J. Biochem. 110:956.
6. Eser S, et al. 2011. Proc Natl Acad Sci USA. 108:9945.
7. Yasukochi A, et al. 2010. Biol Chem. 391: 947. - Gene ID
- 1510 View all products for this Gene ID
- UniProt
- View information about Cathepsin E on UniProt.org
Related FAQs
- Why choose BioLegend recombinant proteins?
-
• Each lot of product is quality-tested for bioactivity as indicated on the data sheet.
• Greater than 95% Purity or higher, tested on every lot of product.
• 100% Satisfaction Guarantee for quality performance, stability, and consistency.
• Ready-to-use liquid format saves time and reduces challenges associated with reconstitution.
• Bulk and customization available. Contact us.
• Learn more about our Recombinant Proteins. - How does the activity of your recombinant proteins compare to competitors?
-
We quality control each and every lot of recombinant protein. Not only do we check its bioactivity, but we also compare it against other commercially available recombinant proteins. We make sure each recombinant protein’s activity is at least as good as or better than the competition’s. In order to provide you with the best possible product, we ensure that our testing process is rigorous and thorough. If you’re curious and eager to make the switch to BioLegend recombinants, contact your sales representative today!
- What is the specific activity or ED50 of my recombinant protein?
-
The specific activity range of the protein is indicated on the product datasheets. Because the exact activity values on a per unit basis can largely fluctuate depending on a number of factors, including the nature of the assay, cell density, age of cells/passage number, culture media used, and end user technique, the specific activity is best defined as a range and we guarantee the specific activity of all our lots will be within the range indicated on the datasheet. Please note this only applies to recombinants labeled for use in bioassays. ELISA standard recombinant proteins are not recommended for bioassay usage as they are not tested for these applications.
- Have your recombinants been tested for stability?
-
Our testing shows that the recombinant proteins are able to withstand room temperature for a week without losing activity. In addition the recombinant proteins were also found to withstand four cycles of freeze and thaw without losing activity.
- Does specific activity of a recombinant protein vary between lots?
-
Specific activity will vary for each lot and for the type of experiment that is done to validate it, but all passed lots will have activity within the established ED50 range for the product and we guarantee that our products will have lot-to-lot consistency. Please conduct an experiment-specific validation to find the optimal ED50 for your system.
- How do you convert activity as an ED50 in ng/ml to a specific activity in Units/mg?
-
Use formula Specific activity (Units/mg) = 10^6/ ED50 (ng/mL)
 Login/Register
Login/Register 









Follow Us