- Clone
- PK136 (See other available formats)
- Regulatory Status
- RUO
- Other Names
- NKR-P1C, NKR-P1B, Ly-55, CD161, CD161b, CD161c
- Isotype
- Mouse IgG2a, κ
- Ave. Rating
- Submit a Review
- Product Citations
- publications
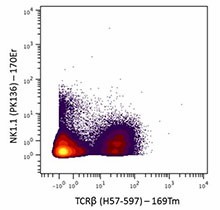
-

Mouse splenocytes stained with 169Tm-anti-TCRβ (H57-597) and 170Er-anti-NK1.1 (PK136). Data provided by DVS Sciences.
| Cat # | Size | Price | Quantity Check Availability | Save | ||
|---|---|---|---|---|---|---|
| 108743 | 100 µg | $106 | ||||
NK-1.1 surface antigen, also known as CD161b/CD161c and Ly-55, is encoded by the NKR-P1B/NKR-P1C gene. It is expressed on NK cells and NK-T cells in some mouse strains, including C57BL/6, FVB/N, and NZB, but not AKR, BALB/c, CBA/J, C3H, DBA/1, DBA/2, NOD, SJL, and 129. Expression of NKR-P1C antigen has been correlated with lysis of tumor cells in vitro and rejection of bone marrow allografts in vivo. NK-1.1 has also been shown to play a role in NK cell activation, IFN-γ production, and cytotoxic granule release. NK-1.1 and DX5 are commonly used as mouse NK cell markers.
Product DetailsProduct Details
- Verified Reactivity
- Mouse
- Antibody Type
- Monoclonal
- Host Species
- Mouse
- Immunogen
- NK-1+ cells from mouse spleen and bone marrow
- Formulation
- Phosphate-buffered solution, pH 7.2, containing 0.09% sodium azide and EDTA.
- Preparation
- The antibody was purified by affinity chromatography.
- Concentration
- 1.0 mg/ml
- Storage & Handling
- The antibody solution should be stored undiluted between 2°C and 8°C.
- Application
-
FC - Quality tested
CyTOF® - Verified - Recommended Usage
-
This product is suitable for use with the Maxpar® Metal Labeling Kits. For metal labeling using Maxpar® Ready antibodies, proceed directly to the step to Partially Reduce the Antibody by adding 100 µl of Maxpar® Ready antibody to 100 µl of 4 mM TCEP-R in a 50 kDa filter and continue with the protocol. Always refer to the latest version of Maxpar® User Guide when conjugating Maxpar® Ready antibodies.
- Application Notes
-
Additional reported applications (for the relevant formats) include: immunoprecipitation1,2, complement-dependent cytotoxicity3, in vivo depletion4,5,9,10, mediation of in vitro redirected lysis6, blocking of NK cell function7, induction of proliferation8, immunohistochemical staining of frozen sections11, immunofluorescence microscopy11, and spatial biology (IBEX)16,17. The LEAF™ purified antibody (Endotoxin <0.1 EU/µg, Azide-Free, 0.2 µm filtered) is recommended for functional assays (Cat. No. 108712).
- Additional Product Notes
-
Maxpar® is a registered trademark of Standard BioTools Inc.
-
Application References
(PubMed link indicates BioLegend citation) -
- Carlyle JR, et al. 1999. J. Immunol. 162:5917. (IP)
- Sentman CL, et al. 1989. Hybridoma 8:605. (IP)
- Koo GC, et al. 1984. Hybridoma 3:301. (Cyt)
- Sentman CL, et al. 1989. J. Immunol. 142:1847. (Deplete)
- Koo GC, et al. 1986. J. Immunol. 137:3742. (Deplete)
- Karlhofer FM, et al. 1991. J. Immunol. 146:3662.
- Kung SK, et al. 1999. J. Immunol. 162:5876. (Block)
- Reichlin A, et al. 1998. Immunol. Cell Biol. 76:143.
- Drobyski W, et al. 1996. Blood 87:5355. (Deplete)
- Andoniou CE, et al. 2005. Nat. Immunol. 6:1011. (Deplete)
- Kanwar JR, et al. 2001. J. Natl. Cancer Inst. 93:1541. (IHC, IF)
- Kroemer A, et al. 2008. J. Immunol. 180:7818. PubMed
- Kim JY, et al. 2009. Exp Mol Med. 30:288. PubMed
- Bankoti J, et al. 2010. Toxicol. Sci. 115:422. (FC) PubMed
- Lee H, et al. 2014. Invest Ophthalmol Vis Sci. 55:2885. PubMed
- Radtke AJ, et al. 2020. Proc Natl Acad Sci U S A. 117:33455-65. (SB) PubMed
- Radtke AJ, et al. 2022. Nat Protoc. 17:378-401. (SB) PubMed
- Product Citations
-
- RRID
-
AB_2562803 (BioLegend Cat. No. 108743)
Antigen Details
- Structure
- NKR-P1 gene family
- Distribution
-
NK and NK-T cells in the NK1.1 mouse strains (C57BL, FVB/N, NZB)
- Function
- NK cell activation, IFN-γ production, cytotoxic granule release
- Cell Type
- NK cells, NKT cells
- Biology Area
- Immunology, Innate Immunity
- Antigen References
-
1. Lanier LL. 1997. Immunity 6:371.
2. Yokoyama WM, et al. 1993. Ann. Rev. Immunol. 11:613.
3. Koo GC, et al. 1986. J. Immunol. 137:3742.
4. Giorda R, et al. 1991. J. Immunol. 147:1701. - Gene ID
- 17059 View all products for this Gene ID
- UniProt
- View information about NK-1.1 on UniProt.org
Related Pages & Pathways
Pages
Related FAQs
- Can I obtain CyTOF data related to your Maxpar® Ready antibody clones?
-
We do not test our antibodies by mass cytometry or on a CyTOF machine in-house. The data displayed on our website is provided by Fluidigm®. Please contact Fluidigm® directly for additional data and further details.
- Can I use Maxpar® Ready format clones for flow cytometry staining?
-
We have not tested the Maxpar® Ready antibodies formulated in solution containing EDTA for flow cytometry staining. While it is likely that this will work in majority of the situations, it is best to use the non-EDTA formulated version of the same clone for flow cytometry testing. The presence of EDTA in some situations might negatively affect staining.
- I am having difficulty observing a signal after conjugating a metal tag to your Maxpar® antibody. Please help troubleshoot.
-
We only supply the antibody and not test that in house. Please contact Fluidigm® directly for troubleshooting advice: http://techsupport.fluidigm.com/
- Is there a difference between buffer formulations related to Maxpar® Ready and purified format antibodies?
-
The Maxpar® Ready format antibody clones are formulated in Phosphate-buffered solution, pH 7.2, containing 0.09% sodium azide and EDTA. The regular purified format clones are formulated in solution that does not contain any EDTA. Both formulations are however without any extra carrier proteins.
Other Formats
View All NK-1.1 Reagents Request Custom ConjugationCompare Data Across All Formats
This data display is provided for general comparisons between formats.
Your actual data may vary due to variations in samples, target cells, instruments and their settings, staining conditions, and other factors.
If you need assistance with selecting the best format contact our expert technical support team.
-
APC anti-mouse NK-1.1

C57BL/6 mouse splenocytes were stained with CD49b (DX5) FITC... 
-
Biotin anti-mouse NK-1.1

C57BL/6 mouse splenocytes were stained with CD49b (DX5) APC ... 
-
FITC anti-mouse NK-1.1

C57BL/6 mouse splenocytes were stained with NK1.1 (clone PK1... -
PE anti-mouse NK-1.1

C57BL/6 mouse splenocytes were stained with CD49b (DX5) APC ... -
Purified anti-mouse NK-1.1

C57BL/6 mouse splenocytes were stained with CD49b (DX5) APC ... -
PE/Cyanine7 anti-mouse NK-1.1

C57BL/6 mouse splenocytes were stained with DX5 APC and NK1.... -
PE/Cyanine5 anti-mouse NK-1.1

C57BL/6 mouse splenocytes were stained with DX5 APC and NK1.... -
Alexa Fluor® 488 anti-mouse NK-1.1

C57BL/6 mouse splenocytes were stained with CD49b (DX5) APC ... -
Alexa Fluor® 647 anti-mouse NK-1.1
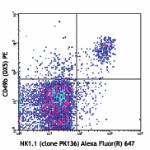
C57BL/6 mouse splenocytes were stained with CD49b (DX5) PE a... 
-
Pacific Blue™ anti-mouse NK-1.1
C57BL/6 splenocytes stained with PK136 Pacific Blue™ -
Brilliant Violet 711™ anti-mouse NK-1.1

C57BL/6 mouse splenocytes were stained with CD49b/DX5 FITC a... 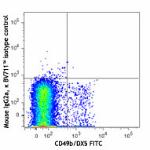
-
APC/Cyanine7 anti-mouse NK-1.1

C57BL/6 mouse splenocytes were stained with CD49b (clone DX5... -
PerCP anti-mouse NK-1.1

C57BL/6 mouse splenocytes were stained with CD49b (DX5) FITC... 
-
PerCP/Cyanine5.5 anti-mouse NK-1.1

C57BL/6 mouse splenocytes were stained with CD49b (pan-NK ce... -
Alexa Fluor® 700 anti-mouse NK-1.1

C57BL/6 splenocytes were stained with CD49b (clone DX5) PE a... -
Brilliant Violet 421™ anti-mouse NK-1.1

C57BL/6 mouse splenocytes were stained with CD49b/DX5 PE and... 
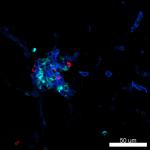
Confocal image of C57BL/6 mouse liver sample acquired using ... 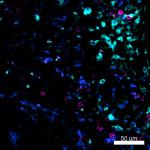
Mice were injected subcutaneously with sheep red blood cells... -
Brilliant Violet 570™ anti-mouse NK-1.1

C57BL/6 mouse splenocytes were stained with CD49b/DX5 FITC a... 
-
Brilliant Violet 650™ anti-mouse NK-1.1
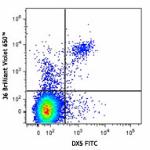
C57BL/6 mouse splenocytes were stained with NK1.1 (clone PK1... -
Brilliant Violet 510™ anti-mouse NK-1.1

C57BL/6 mouse splenocytes were stained with CD49b/DX5 APC an... 
-
Brilliant Violet 605™ anti-mouse NK-1.1

C57BL/6 mouse splenocytes were stained with CD49b/DX5 APC an... 
-
Purified anti-mouse NK-1.1 (Maxpar® Ready)

Mouse splenocytes stained with 169Tm-anti-TCRβ (H57-597) and... -
PE/Dazzle™ 594 anti-mouse NK-1.1

C57BL/6 mouse splenocytes were stained with CD49b FITC and N... 
-
Brilliant Violet 785™ anti-mouse NK-1.1

C57BL/6 mouse splenocytes were stained with CD49b/DX5 FITC a... 
-
APC/Fire™ 750 anti-mouse NK-1.1

C57BL/6 mouse splenocytes were stained with CD49b FITC and N... 
-
TotalSeq™-A0118 anti-mouse NK-1.1
-
Ultra-LEAF™ Purified anti-mouse NK-1.1

C57BL/6 mouse splenocytes were stained with CD49b (DX5) APC ... -
TotalSeq™-B0118 anti-mouse NK-1.1
-
TotalSeq™-C0118 anti-mouse NK-1.1
-
PE/Fire™ 810 anti-mouse NK-1.1

C57BL/6 mouse splenocytes were stained with anti-mouse CD49b... -
PE/Fire™ 640 anti-mouse NK-1.1

C57BL/6 mouse splenocytes were stained with anti-mouse CD49b... -
Spark UV™ 387 anti-mouse NK-1.1

C57BL/6 mouse splenocytes were stained with anti-mouse CD49d... -
PE/Fire™ 700 anti-mouse NK-1.1

C57BL/6 mouse splenocytes were stained with anti-mouse CD49b...
 Login/Register
Login/Register 












Follow Us