- Clone
- UC7-13D5 (See other available formats)
- Regulatory Status
- RUO
- Other Names
- TCR-γ/δ, γ/δ TCR
- Isotype
- Armenian Hamster IgG
- Ave. Rating
- Submit a Review
- Product Citations
- publications
-

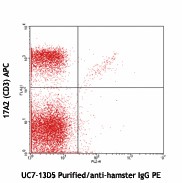
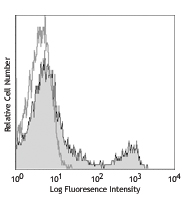
C57BL/6 splenocytes stained with 17A2 (CD3) APC and purified UC7-13D5, followed by anti-Armenian hamster IgG PE
T cell receptor (TCR) is a heterodimer consisting of an α and a β chain (TCR α/β) or a γ and a δ chain (TCR γ/δ). TCR γ/δ belongs to the immunoglobulin superfamily, involved in the recognition of certain bacterial and tumor antigens bound to MHC class I. The TCR γ/δ associates with CD3 and is expressed on a T cell subset found in the thymus, the intestinal epithelium, and the peripheral lymphoid tissues and peritoneum. Most γ/δ T cells are CD4-/CD8-, some are CD8+. T cells expressing the TCR γ/δ have been shown to play a role in oral tolerance, tumor-associated tolerance, and autoimmune disease. It has been reported that γ/δ T cells also play a principal role in antigen presentation. Immobilized UC7-13D5 antibody has been reported to activate TCR-γ/δ-bearing T cells in vitro, and to deplete peripheral TCR-γ/δ-bearing T cells in vivo.
Product DetailsProduct Details
- Verified Reactivity
- Mouse
- Antibody Type
- Monoclonal
- Host Species
- Armenian Hamster
- Formulation
- 0.2 µm filtered in phosphate-buffered solution, pH 7.2, containing no preservative.
- Endotoxin Level
- Less than 0.01 EU/µg of the protein (< 0.001 ng/µg of the protein) as determined by the LAL test.
- Preparation
- The Ultra-LEAF™ (Low Endotoxin, Azide-Free) antibody was purified by affinity chromatography.
- Concentration
- The antibody is bottled at the concentration indicated on the vial, typically between 2 mg/mL and 3 mg/mL. Older lots may have also been bottled at 1 mg/mL. To obtain lot-specific concentration and expiration, please enter the lot number in our Certificate of Analysis online tool.
- Storage & Handling
- The antibody solution should be stored undiluted between 2°C and 8°C. This Ultra-LEAF™ solution contains no preservative; handle under aseptic conditions.
- Application
-
FC - Quality tested
IP, Stim, Depletion - Reported in the literature, not verified in house - Recommended Usage
-
Each lot of this antibody is quality control tested by immunofluorescent staining with flow cytometric analysis. For flow cytometric staining, the suggested use of this reagent is ≤ 1.0 µg per million cells in 100 µl volume or 100 µl of whole blood. It is recommended that the reagent be titrated for optimal performance for each application.
- Application Notes
-
Additional reported applications (for the relevant formats) include: immunoprecipitation1, in vitro stimulation1,2,4. Although several papers have reported in vivo depletion3, 5,6, another study found that the effect of the antibody could be the functional blocking of the TCR signaling, rather than cell depletion7. The Ultra-LEAF™ purified antibody (Endotoxin < 0.01 EU/µg, Azide-Free, 0.2 µm filtered) is recommended for functional assays (Cat. Nos. 107515 - 107520).
Though clone UC7-13D5 has been used for the depletion of TCR γ/δ T cells in vivo3-5, there is a report suggesting that binding of clone UC7-13D5 to the γ/δ TCR leads to downregulation of the γ/δ TCR, making the γ/δ T cells undetectable although they are still present7. -
Application References
(PubMed link indicates BioLegend citation) -
- Bluestone JA, et al. 1991. Immunol. Rev. 120:5. (IP Costim)
- Kruisbeek AM, et al. 1991. In Current Protocols in Immunology p. 3.12.1 - 3.12.14. (Costim)
- Hiromatsu K, et al. 1992. J. Exp. Med. 175:49. (Deplete)
- Dieli F, et al. 1997. Eur. J. Immunol. 27:206. (Costim)
- van der Heyde HC, et al. 1995. J. Immunol. 154:3985. (Deplete)
- Pollinger B, et al. 2011. J. Immunol. 186:2602. PubMed
- Koenecke C, et al. 2009. Eur J Immunol. 39(2):372-9.
- Product Citations
-
- RRID
-
AB_2813962 (BioLegend Cat. No. 107515)
AB_2813963 (BioLegend Cat. No. 107516)
AB_2813964 (BioLegend Cat. No. 107517)
AB_2813965 (BioLegend Cat. No. 107518)
AB_2813966 (BioLegend Cat. No. 107519)
AB_2813967 (BioLegend Cat. No. 107520)
Antigen Details
- Structure
- Ig superfamily, associates with CD3 complex
- Distribution
-
T subset in thymus, intestinal epithelium, peripheral lymphoid tissues and peritoneum
- Function
- Antigen recognition
- Ligand/Receptor
- Some bacterial or tumor antigens bound MHC class I
- Cell Type
- Epithelial cells, T cells, Tregs
- Biology Area
- Adaptive Immunity, Immunology
- Molecular Family
- TCRs
- Antigen References
-
1. Skarstein K, et al. 1995. Immunology 81:497.
2. Harrison LC, et al. 1996. J. Exp. Med. 184:2167.
3. Wildner G, et al. 1996. Eur. J. Immunol. 26:2140.
4. Brandes M, et al. 2005. Science 309:264. - Gene ID
- 110066 View all products for this Gene ID 110067 View all products for this Gene ID
- UniProt
- View information about TCR gamma/delta on UniProt.org
Related FAQs
- Do you guarantee that your antibodies are totally pathogen free?
-
BioLegend does not test for pathogens in-house aside from the GoInVivo™ product line. However, upon request, this can be tested on a custom basis with an outside, independent laboratory.
- Does BioLegend test each Ultra-LEAF™ antibody by functional assay?
-
No, BioLegend does not test Ultra-LEAF™ antibodies by functional assays unless otherwise indicated. Due to the possible complexities and variations of uses of biofunctional antibodies in different assays and because of the large product portfolio, BioLegend does not currently perform functional assays as a routine QC for the antibodies. However, we do provide references in which the antibodies were used for functional assays and we do perform QC to verify the specificity and quality of the antibody based on our strict specification criteria.
- Does BioLegend test each Ultra-LEAF™ antibody for potential pathogens?
-
No, BioLegend does not test for pathogens in-house unless otherwise indicated. However, we can recommend an outside vendor to perform this testing as needed.
- Have you tested this Ultra-LEAF™ antibody for in vivo or in vitro applications?
-
We don't test our antibodies for in vivo or in vitro applications unless otherwise indicated. Depending on the product, the TDS may describe literature supporting usage of a particular product for bioassay. It may be best to further consult the literature to find clone specific information.
Other Formats
View All TCR γ/δ Reagents Request Custom Conjugation| Description | Clone | Applications |
|---|---|---|
| FITC anti-mouse TCR γ/δ | UC7-13D5 | FC |
| Purified anti-mouse TCR γ/δ | UC7-13D5 | FC,IP,Stim,Depletion |
| PE anti-mouse TCR γ/δ | UC7-13D5 | FC |
| Alexa Fluor® 488 anti-mouse TCR γ/δ | UC7-13D5 | FC |
| Ultra-LEAF™ Purified anti-mouse TCR γ/δ | UC7-13D5 | FC,IP,Stim,Depletion |
| TotalSeq™-C0986 anti-mouse TCR γ/δ | UC7-13D5 | PG |
Customers Also Purchased
Compare Data Across All Formats
This data display is provided for general comparisons between formats.
Your actual data may vary due to variations in samples, target cells, instruments and their settings, staining conditions, and other factors.
If you need assistance with selecting the best format contact our expert technical support team.
-
FITC anti-mouse TCR γ/δ

C57BL/6 splenocytes stained with CD3 (145-2C11) APC and UC7-... -
Purified anti-mouse TCR γ/δ

C57BL/6 splenocytes stained with 17A2 (CD3) APC and purified... -
PE anti-mouse TCR γ/δ
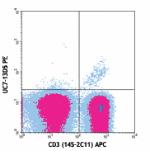
C57BL/6 mouse lymph node cells stained with 145-2C11 APC and... -
Alexa Fluor® 488 anti-mouse TCR γ/δ
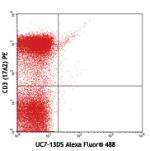
C57BL/6 mouse splenocytes stained with CD3 (17A2) PE and UC7... -
Ultra-LEAF™ Purified anti-mouse TCR γ/δ

C57BL/6 splenocytes stained with 17A2 (CD3) APC and purified... -
TotalSeq™-C0986 anti-mouse TCR γ/δ
 Login / Register
Login / Register 








Follow Us