- Regulatory Status
- RUO
- Other Names
- Ciliary neurotrophic factor
- Ave. Rating
- Submit a Review
- Product Citations
- publications
-
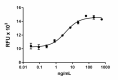
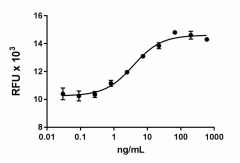
Rat CNTF induces the proliferation of TF-1 human erythroleukemic cells in a dose dependent manner. ED50 for this effect is 3-18 ng/mL. -
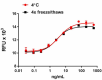
Stability testing for rat CNTF. Recombinant rat CNTF was aliquoted in 20 mM Tris, 150 mM NaCl, 1 mM EDTA, 2 mM CHAPS, 2 mM TCEP, pH 7.5 at 0.2 mg/mL and one aliquot was kept at 4°C (control), and another was frozen and thawed four times (4x freeze/thaws). After this procedure, the samples were tested for their ability to induce TF-1 cell proliferation.
CNTF, ciliary neurotrophic factor, was initially identified as a survival factor for cultured embryonic chick parasympathetic neurons. The rat protein has 84% similarity to the human and rabbit proteins. It lacks an N-terminal signal sequence and consensus sequences for glycosylation or hydrophobic regions. The CNTF profile shows similar sequence and structure motifs to hematopoietic cytokines (IL-6, IL-11, LIF, oncostatin M, cardiotrophin-1) and thus was considered a member of the IL-6 family. CNTF exerts effects on glial cells and neurons; CNTF activates astrocytes, which promote their capacity to support neurons and oligodendroglia. CNTF stimulates astrocytes to secrete FGF-2 and rat microglia to secrete glial cell line-derived neurotrophic factor (GDNF). CNTF is produced by astrocytes after brain injury. The CNTF receptor complex includes the CNTF binding protein (CNTFRα), and two signaling chains leukemia inhibitory factor receptor β (LIFRβ) and the signal transducer of IL-6 (gp130). CNTFRα does not have transmembrane or intracellular domains, which means it does not induce signal transduction per se. CNTFRα is associated to the cell membrane via glycosyl phosphatidylinositol linkage, and it can be released by phospholipase C to produce a soluble receptor. The complex formed by CNTFRα and CNTF serve as agonists for cells that do not express CNTFRα. In addition, human CNTF can recruit and activate another receptor in this case CNTFRα is substituted by IL-6Rα. Cardiotrophin-like cytokine (CLC) also binds to CNTFRα, which activates the gp130 and LIFRb subunits. CNTF reduces the symptoms of experimental autoimmune encephalomyelitis, and its absence exacerbates the severity of multiple sclerosis disability. CNTF has been studied in retinal degenerative disorders and was shown to have a potential clinical application.
Product DetailsProduct Details
- Source
- Rat CNTF amino acids Met- (Ala2 - Met200) (Accession #P20294.1) was expressed in E. coli.
- Molecular Mass
- The 200 amino acid recombinant protein has a predicted molecular mass of approximately 22.85 kD. The protein migrates approximately at 23 kD in DTT-reducing and non-reducing conditions by SDS-PAGE. The predicted N-terminal amino acid is Met.
- Purity
- > 98% by SDS-PAGE gel and HPLC analyses.
- Formulation
- 0.22 µm filtered protein solution is in 20 mM Tris, 150 mM NaCl, 1 mM EDTA, 2 mM CHAPS, 2 mM TCEP, pH 7.5.
- Endotoxin Level
- Less than 0.1 EU per µg cytokine as determined by the LAL method.
- Concentration
- 10 and 25 µg sizes are bottled at 200 µg/mL. 100 µg size and larger sizes are lot-specific and bottled at the concentration indicated on the vial. To obtain lot-specific concentration and expiration, please enter the lot number in our Certificate of Analysis online tool.
- Storage & Handling
- Unopened vial can be stored between 2°C and 8°C for up to 2 weeks, at -20°C for up to six months, or at -70°C or colder until the expiration date. For maximum results, quick spin vial prior to opening. The protein can be aliquoted and stored at -20°C or colder. Stock solutions can also be prepared at 50 - 100 µg/mL in appropriate sterile buffer, carrier protein such as 0.2 - 1% BSA or HSA can be added when preparing the stock solution. Aliquots can be stored between 2°C and 8°C for up to one week and stored at -20°C or colder for up to 3 months. Avoid repeated freeze/thaw cycles.
- Activity
- Rat CNTF induces the proliferation of TF-1 human erythroleukemic cells in a dose dependent manner. The ED50 is 3 - 18 ng/mL.
- Application
-
Bioassay
- Application Notes
-
BioLegend carrier-free recombinant proteins provided in liquid format are shipped on blue ice. Our comparison testing data indicates that when handled and stored as recommended, the liquid format has equal or better stability and shelf-life compared to commercially available lyophilized proteins after reconstitution. Our liquid proteins are verified in-house to maintain activity after shipping on blue ice and are backed by our 100% satisfaction guarantee. If you have any concerns, contact us at tech@biolegend.com.
Antigen Details
- Structure
- Dimer
- Distribution
-
Astrocytes, microglia, hepatocytes, skeletal muscle, embryonic stem cells, and bone marrow stromal cells.
- Function
- Stimulates gene expression, along with cell survival or differentiation in neuronal cell types. It has a potent trophic factor for motor neurons. IL-1b, TNFα (TNF-α), and EGF induces the release of CNTF from astrocytes.
- Interaction
- Sensory, sympathethic, ciliary, motor neurons, microglia, retinal ganglion cells, adipocytes, and skeletal muscle cells.
- Ligand/Receptor
- Trimeric complex form by CNTF receptor-α (CNTFRα), LIF receptor (LIFR), and IL-6Rα (CD126).
- Cell Type
- Neural Stem Cells, Hematopoietic stem and progenitors, Embryonic Stem Cells
- Biology Area
- Cell Biology, Stem Cells
- Molecular Family
- Growth Factors, Cytokines/Chemokines
- Antigen References
-
- Negro A, et al. 1991. Eur. J. Biochem. 201:289.
- Bazan JF. 1991. Neuron. 7:197.
- Davis S, et al. 1993. Science. 260:1805.
- Plun-Favreau H, et al. 2001. EMBO J. 20:1692.
- Lin HW, et al. 2009. J. Neuroinflammation. 6:7.
- Wen R, et al. 2012. Prog. Retin. Eye Res. 31:136.
- Tormo AJ, et al. 2012. Cytokine. 60:653.
- Gene ID
- 25707 View all products for this Gene ID
- UniProt
- View information about CNTF on UniProt.org
Related FAQs
- Why choose BioLegend recombinant proteins?
-
• Each lot of product is quality-tested for bioactivity as indicated on the data sheet.
• Greater than 95% Purity or higher, tested on every lot of product.
• 100% Satisfaction Guarantee for quality performance, stability, and consistency.
• Ready-to-use liquid format saves time and reduces challenges associated with reconstitution.
• Bulk and customization available. Contact us.
• Learn more about our Recombinant Proteins. - How does the activity of your recombinant proteins compare to competitors?
-
We quality control each and every lot of recombinant protein. Not only do we check its bioactivity, but we also compare it against other commercially available recombinant proteins. We make sure each recombinant protein’s activity is at least as good as or better than the competition’s. In order to provide you with the best possible product, we ensure that our testing process is rigorous and thorough. If you’re curious and eager to make the switch to BioLegend recombinants, contact your sales representative today!
- What is the specific activity or ED50 of my recombinant protein?
-
The specific activity range of the protein is indicated on the product datasheets. Because the exact activity values on a per unit basis can largely fluctuate depending on a number of factors, including the nature of the assay, cell density, age of cells/passage number, culture media used, and end user technique, the specific activity is best defined as a range and we guarantee the specific activity of all our lots will be within the range indicated on the datasheet. Please note this only applies to recombinants labeled for use in bioassays. ELISA standard recombinant proteins are not recommended for bioassay usage as they are not tested for these applications.
- Have your recombinants been tested for stability?
-
Our testing shows that the recombinant proteins are able to withstand room temperature for a week without losing activity. In addition the recombinant proteins were also found to withstand four cycles of freeze and thaw without losing activity.
- Does specific activity of a recombinant protein vary between lots?
-
Specific activity will vary for each lot and for the type of experiment that is done to validate it, but all passed lots will have activity within the established ED50 range for the product and we guarantee that our products will have lot-to-lot consistency. Please conduct an experiment-specific validation to find the optimal ED50 for your system.
- How do you convert activity as an ED50 in ng/ml to a specific activity in Units/mg?
-
Use formula Specific activity (Units/mg) = 10^6/ ED50 (ng/mL)
 Login / Register
Login / Register 








Follow Us