- Regulatory Status
- RUO
- Other Names
- AP-TNAP, TNAP, Tissue-Nonspecific ALP, APTNAP, Alkaline Phosphatase, Biomineralization Associated; Alkaline Phosphatase, Tissue-Nonspecific Enzyme (TNAP); Alkaline Phosphatase, Liver/Bone/Kidney Isozyme
- Ave. Rating
- Submit a Review
- Product Citations
- publications
-
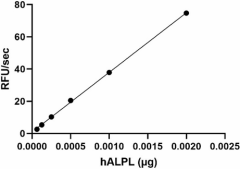
Recombinant human ALPL activity is measured by its ability to cleave the fluorogenic substrate, 4-MUP. The increase of the product was monitored by an increase in intensity of fluorescence at 445 nm with excitation at 365 nm. The specific activity is > 180,000 pmol/min/µg in the presence of 0.0005 µg of recombinant human ALPL. -
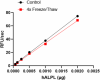
Stability Testing for Recombinant Human ALPL. Recombinant human ALPL was aliquoted in 50 mM HEPES, 100 mM NaCl, 1 mM MgCl2, 0.1 mM ZnCl2, pH 7. One aliquot was frozen and thawed four times (4x Freeze/Thaw) and compared to the control that was kept at 4⁰C (Control). The samples were tested for their ability to cleave the fluorogenic substrate, 4-MUP. The increase of the product was monitored by an increase in intensity of fluorescence at 445 nm with excitation at 365 nm. The specific activity is > 180,000 pmol/min/µg in the presence of 0.0005 µg of recombinant human ALPL.
| Cat # | Size | Price | Quantity Check Availability | Save | ||
|---|---|---|---|---|---|---|
| 794504 | 25 µg | 184€ | ||||
| 794506 | 100 µg | 540€ | ||||
ALPL is the tissue-nonspecific member of the alkaline phosphatase protein family, which includes four members: the tissue-nonspecific (liver/bone/kidney) ALP also called TNSALP, encoded by ALPL gene, and three tissue-specific ALPs that include intestinal (IAP), placental (PLAP-1), and placental-like 2 or germ cell (GCAP) ALPs; these enzymes are encoded by ALPI, ALPP, and ALPPL2 genes respectively. Both ALPL and ALPI are expressed in many vertebrate species, while PLAP-1 and GCAP are expressed only in mammalians. ALPL catalyzes the dephosphorylation of compounds in many transphosphorylation reactions. ALPL hydrolyze monophosphated esters such as phosphoethanolamine, inorganic pyrophosphate, and pyridoxal 5′-phosphate. ALPL is a homodimeric protein and each monomer possesses 524 amino acids. It is expressed mainly in liver, bone, and kidney tissues, although it can be found in all tissues, typically in a membrane bound form. Its catalytic site harbors two Zn ions and one Mg ion essential for enzyme activity. Mutations in the ALPL gene causing deficiency in ALPL activity result in hypophosphatasia and skeletal defects. ALPL regulates senescence and differentiation in mesenchymal stem cells from bone marrow. ALPL deficiency induces bone mass loss and increase in marrow fat, characteristics of premature bone ageing. ALPL was found to regulate cancer cell motility in some ovarian cancers via a non-canonical WNT pathway.
Product DetailsProduct Details
- Source
- Human ALPL, amino acid (Leu18 - Ser502) (Accession: NM_00478) was expressed in CHO cells. The C-terminus contains 6His-tag.
- Molecular Mass
- The 491 amino acid recombinant protein has a predicted molecular mass of approximately 54.2 kD. The DTT-reduced and non reduced protein migrates at approximately 70-75 kD by SDS-PAGE. The C-terminus contains 6His tag. The predicted N-terminal amino acid is Leu.
- Purity
- > 95%, as determined by Coomassie stained SDS-PAGE
- Formulation
- 0.22 µm filtered protein solution is in 50 mM HEPES, 100 mM NaCl, 1 mM MgCl2, 0.1 mM ZnCl2, pH 7.0.
- Endotoxin Level
- Less than 0.1 EU per µg cytokine as determined by the LAL method
- Concentration
- 25 µg size is bottled at 200 µg/mL. 100 µg size and larger sizes are lot-specific and bottled at the concentration indicated on the vial. To obtain lot-specific concentration and expiration, please enter the lot number in our Certificate of Analysis online tool.
- Storage & Handling
- Unopened vial can be stored at -20°C or -70°C for six months. For maximum results, quick spin vial prior to opening. Avoid repeated freeze/thaw cycles.
- Activity
- Human tissue-nonspecific alkaline phosphatase (ALPL) cleaves the fluorogenic peptide substrate, 4-methylumbelliferyl phosphate (4-MUP). The specific activity is > 180,000 pmol/min/µg in the presence of 0.0005 µg of recombinant human ALPL.
- Application
-
Bioassay
- Additional Product Notes
-
Human ALPL Enzymatic Assay
Human tissue-nonspecific alkaline phosphatase (ALPL) cleaves the fluorogenic peptide substrate, 4-Methylumbelliferyl phosphate (4-MUP). The specific activity is > 180,000 pmol/min/µg in the presence of 0.0005 µg of recombinant human ALPL.- Assay Buffer: 50 mM Tris, 1 mM MgCl2, pH 9.0
- hALPL Substrate: (Sigma, Cat. No. M8883), 25 mg/mL in diH2O
- Recombinant human ALPL
- F16 Black Maxisorp Plate (Nunc, Cat. No 475515)
- Fluorescent plate reader (Model: SpectraMax M3 by Molecular Devices) or equivalent
Procedure
- Dilute the recombinant human ALPL to 0.01 ng/µL in assay buffer.
- Dilute the substrate to 50 µM in assay buffer.
- In a plate load 50 µL of the diluted human ALPL to the wells. Include a substrate blank of 50 µL assay buffer and 50 µL of diluted substrate.
- Start the reaction by adding 50 µL of 50 µM substrate to the wells.
- Read at excitation and emission wavelengths of 365 nm and 445 nm (top read) respectively in kinetic mode for 5 minutes.
- Calculate specific activity:
Specific Activity (pmol/min/µg) = Adjusted Vmax* (RFU/min) x Conversion Factor** (pmol/RFU)
amount of enzyme (µg)*Adjusted for substrate blank
**Derived using calibration standard 4-Methumbelliferone (Sigma, Cat. No. M1381)
Per Well:
Recombinant human ALPL: 0.0005 µg
Substrate: 25 µM
BioLegend carrier-free recombinant proteins provided in liquid format are shipped on blue-ice. Our comparison testing data indicates that when handled and stored as recommended, the liquid format has equal or better stability and shelf-life compared to commercially available lyophilized proteins after reconstitution. Our liquid proteins are verified in-house to maintain activity after shipping on blue ice and are backed by our 100% satisfaction guarantee. If you have any concerns, contact us at tech@biolegend.com.
Antigen Details
- Structure
- Homodimeric enzyme
- Distribution
-
Osteoblast, bone marrow mesenchymal stem cells, liver, bone, kidney
- Function
- Bone and tooth mineralization, metabolism of nucleotides, sequentially hydrolyzes ATP, ADP and AMP
- Interaction
- Monoesters of phosphoric acid, inorganic pyrophosphate, phosphoethanolamine, and pyridoxal 5′-phosphate. Interacts with the ECM, collagen.
- Bioactivity
- Human ALPL activity is measured by its ability to cleave 4-Methylumbelliferyl phosphate (4-MUP). The accumulation of cleavage product is quantified by the increase in fluorescence at excitation and emission of 365 and 455 nm, respectively.
- Cell Type
- Embryonic Stem Cells, Mesenchymal Stem Cells, Neutrophils, Osteoblasts
- Biology Area
- Cell Biology, Immuno-Oncology, Stem Cells
- Molecular Family
- Enzymes and Regulators, Proteases
- Antigen References
-
- Weis MJ, et al. 1988. J Biol Chem. 263:12002.
- Kim EE and Wyckoff HW. 1991. J Mol Biol. 218:449.
- Mornet E. 2000. Human Mutation. 15:309.
- Yang Y, et al. 2012. Front Immunol. 3:314.
- Silvent J, et al. 2014. J Biol Chem. 289:24168.
- Liu W, et al. 2018. Bone Res. 6:27.
- Lou M, et al. 2019. Biochem Biophys Res Commun. 513:528.
- Gene ID
- 249 View all products for this Gene ID
- UniProt
- View information about ALPL on UniProt.org
Related FAQs
- Why choose BioLegend recombinant proteins?
-
• Each lot of product is quality-tested for bioactivity as indicated on the data sheet.
• Greater than 95% Purity or higher, tested on every lot of product.
• 100% Satisfaction Guarantee for quality performance, stability, and consistency.
• Ready-to-use liquid format saves time and reduces challenges associated with reconstitution.
• Bulk and customization available. Contact us.
• Learn more about our Recombinant Proteins. - How does the activity of your recombinant proteins compare to competitors?
-
We quality control each and every lot of recombinant protein. Not only do we check its bioactivity, but we also compare it against other commercially available recombinant proteins. We make sure each recombinant protein’s activity is at least as good as or better than the competition’s. In order to provide you with the best possible product, we ensure that our testing process is rigorous and thorough. If you’re curious and eager to make the switch to BioLegend recombinants, contact your sales representative today!
- What is the specific activity or ED50 of my recombinant protein?
-
The specific activity range of the protein is indicated on the product datasheets. Because the exact activity values on a per unit basis can largely fluctuate depending on a number of factors, including the nature of the assay, cell density, age of cells/passage number, culture media used, and end user technique, the specific activity is best defined as a range and we guarantee the specific activity of all our lots will be within the range indicated on the datasheet. Please note this only applies to recombinants labeled for use in bioassays. ELISA standard recombinant proteins are not recommended for bioassay usage as they are not tested for these applications.
- Have your recombinants been tested for stability?
-
Our testing shows that the recombinant proteins are able to withstand room temperature for a week without losing activity. In addition the recombinant proteins were also found to withstand four cycles of freeze and thaw without losing activity.
- Does specific activity of a recombinant protein vary between lots?
-
Specific activity will vary for each lot and for the type of experiment that is done to validate it, but all passed lots will have activity within the established ED50 range for the product and we guarantee that our products will have lot-to-lot consistency. Please conduct an experiment-specific validation to find the optimal ED50 for your system.
- How do you convert activity as an ED50 in ng/ml to a specific activity in Units/mg?
-
Use formula Specific activity (Units/mg) = 10^6/ ED50 (ng/mL)
 Login / Register
Login / Register 








Follow Us